Abundant supplies of fresh water are essential to the development of industry. Enormous quantities are required for the cooling of products and equipment, for process needs, for boiler feed, and for sanitary and potable water supply.
THE PLANETARY WATER CYCLE
Industry is a small participant in the global water cycle .The finite amount of water on the planet participates in a very complicated recycling scheme that provides for its reuse. This recycling of water is termed the "Hydrologic Cycle" (see Figure 1-1).
Evaporation under the influence of sunlight takes water from a liquid to a gaseous phase. The water may condense in clouds as the temperature drops in the upper atmosphere. Wind transports the water over great distances before releasing it in some form of precipitation. As the water condenses and falls to the ground, it absorbs gases from the environment. This is the principal cause of acid rain and acid snow.
WATER AS A SOLVENT
Pure water (H20) is colorless, tasteless, and odorless. It is composed of hydrogen and oxygen. Because water becomes contaminated by the substances with which it comes into contact, it is not available for use in its pure state. To some degree, water can dissolve every naturally occurring substance on the earth. Because of this property, water has been termed a "universal solvent." Although beneficial to mankind, the solvency power of water can pose a major threat to industrial equipment. Corrosion reactions cause the slow dissolution of metals by water. Deposition reactions, which produce scale on heat transfer surfaces, represent a change in the solvency power of water as its temperature is varied. The control of corrosion and scale is a major focus of water treatment technology.
WATER IMPURITIES
Water impurities include dissolved and suspended solids. Calcium bicarbonate is a soluble salt. A solution of calcium bicarbonate is clear, because the calcium and bicarbonate are present as atomic sized ions which are not large enough to reflect light. Some soluble minerals impart a color to the solution. Soluble iron salts produce pale yellow or green solutions; some copper salts form intensely blue solutions. Although colored, these solutions are clear. Suspended solids are substances that are not completely soluble in water and are present as particles. These particles usually impart a visible turbidity to the water. Dissolved and suspended solids are present in most surface waters. Seawater is very high in soluble sodium chloride; suspended sand and silt make it slightly cloudy. An extensive list of soluble and suspended impurities found in water is given in Table 1-1.
Table 1-1. Common impurities found in fresh water.
|
Constituent
|
Chemical Formula
|
Difficulties Caused
|
Means of Treatment
|
|||||
| Turbidity | non-expressed in analysis as units | imparts unsightly appearance to water; deposits in water lines, process equipment, etc.; interferes with most process uses | coagulation, settling, and filtration | |||||
| Hardness | calcium and magnesium salts, expressed as CaCO3 | chief source of scale in heat exchange equipment, boilers, pipe lines, etc.; forms curds with soap, interferes with dyeing, etc. | softening; demineralization; internal boiler water treatment; surface active agents | |||||
| Alkalinity |
bicarbonate(HCO3-), carbonate (CO32-), and hydroxide(OH-), expressed as CaCO3 |
foam and carryover of solids with steam; embrittlement of boiler steel; bicarbonate and carbonate produce CO2 in steam, a source of corrosion in condensate lines | lime and lime-soda softening; acid treatment; hydrogen zeolite softening; demineralization dealkalization by anion exchange | |||||
| Free Mineral Acid | H2SO4 , HCI. etc., expressed as CaCO3 | corrosion | neutralization with alkalies | |||||
| Carbon Dioxide | CO2 | corrosion in water lines, particularly steam and condensate lines | aeration, deaeration, neutralization with alkalies | |||||
| PH | hydrogen ion concentration defined as:
|
pH varies according to acidic or alkaline solids in water; most natural waters have a pH of 6.0-8.0 | pH can be increased by alkalies and decreased by acids | |||||
| Sulfate | SO42- | adds to solids content of water, but in itself is not usually significant, combines with calcium to form calcium sulfate scale | demineralization, reverse osmosis, electrodialysis, evaporation | |||||
| Chloride | Cl - | adds to solids content and increases corrosive character of water | demineralization, reverse osmosis, electrodialysis, evaporation | |||||
| Nitrate | NO3- | adds to solids content, but is not usually significant industrially: high concentrations cause methemoglobinemia in infants; useful for control of boiler metal embrittlement | demineralization, reverse osmosis, electrodialysis, evaporation | |||||
| Fluoride | F- | cause of mottled enamel in teeth; also used for control of dental decay: not usually significant industrially | adsorption with magnesium hydroxide, calcium phosphate, or bone black; alum coagulation | |||||
| Sodium | Na+ | adds to solids content of water: when combined with OH-, causes corrosion in boilers under certain conditions | demineralization, reverse osmosis, electrodialysis, evaporation | |||||
| Silica | SiO2 | scale in boilers and cooling water systems; insoluble turbine blade deposits due to silica vaporization | hot and warm process removal by magnesium salts; adsorption by highly basic anion exchange resins, in conjunction with demineralization, reverse osmosis, evaporation | |||||
| Iron | Fe2+ (ferrous) Fe3+ (ferric) |
discolors water on precipitation; source of deposits in water lines, boilers. etc.; interferes with dyeing, tanning, papermaking, etc. | aeration; coagulation and filtration; lime softening; cation exchange; contact filtration; surface active agents for iron retention | |||||
| Manganese | Mn2+ | same as iron | same as iron | |||||
| Aluminum | AI3+ | usually present as a result of floc carryover from clarifier; can cause deposits in cooling systems and contribute to complex boiler scales | improved clarifier and filter operation | |||||
| Oxygen | O2 | corrosion of water lines, heat exchange equipment, boilers, return lines, etc. | deaeration; sodium sulfite; corrosion inhibitors | |||||
| Hydrogen Sulfide | H2S | cause of "rotten egg" odor; corrosion | aeration; chlorination; highly basic anion exchange | |||||
| Ammonia | NH3 | corrosion of copper and zinc alloys by formation of complex soluble ion | cation exchange with hydrogen zeolite; chlorination; deaeration | |||||
| Dissolved Solids | none | refers to total amount of dissolved matter, determined by evaporation; high concentrations are objectionable because of process interference and as a cause of foaming in boilers | lime softening and cation exchange by hydrogen zeolite; demineralization, reverse osmosis, electrodialysis, evaporation | |||||
| Suspended Solids | none | refers to the measure of undissolved matter, determined gravimetrically; deposits in heat exchange equipment, boilers, water lines, etc. | subsidence; filtration, usually preceded by coagulation and settling | |||||
| Total Solids | none | refers to the sum of dissolved and suspended solids, determined gravimetrically | see "Dissolved Solids" and "Suspended Solids" |
Surface Water
The ultimate course of rain or melting snow depends on the nature of the terrain over which it flows. In areas consisting of hard packed clay, very little water penetrates the ground. In these cases, the water generates "runoff". The runoff collects in streams and rivers. The rivers empty into bays and estuaries, and the water ultimately returns to the sea, completing one major phase of the hydrologic cycle shown in see Figure 1-1.
As water runs off along the surface, it stirs up and suspends particles of sand and soil, creating silt in the surface water. In addition, the streaming action erodes rocky surfaces, producing more sand. As the surface water cascades over rocks, it is aerated. The combination of oxygen, inorganic nutrients leached from the terrain, and sunlight supports a wide variety of life forms in the water, including algae, fungi, bacteria, small crustaceans, and fish.
Often, river beds are lined with trees, and drainage areas feeding the rivers are forested. Leaves and pine needles constitute a large percentage of the biological content of the water. After it dissolves in the water, this material becomes a major cause of fouling of ion exchange resin used in water treatment.
The physical and chemical characteristics of surface water contamination vary considerably over time. A sudden storm can cause a dramatic short term change in the composition of a water supply. Over a longer time period, surface water chemistry varies with the seasons. During periods of high rainfall, high runoff occurs. This can have a favorable or unfavorable impact on the characteristics of the water, depending on the geochemistry and biology of the terrain.
Surface water chemistry also varies over multi year or multidecade cycles of drought and rainfall. Extended periods of drought severely affect the availability of water for industrial use. Where rivers discharge into the ocean, the incursion of salt water up the river during periods of drought presents additional problems. Industrial users must take surface water variability into account when designing water treatment plants and programs.
Groundwater
Water that falls on porous terrains, such as sand or sandy loam, drains or percolates into the ground. In these cases, the water encounters a wide variety of mineral species arranged in complex layers, or strata. The minerals may include granite, gneiss, basalt, and shale. In some cases, there may be a layer of very permeable sand beneath impermeable clay. Water often follows a complex three dimensional path in the ground. The science of groundwater hydrology involves the tracking of these water movements.
Table 1-2. A comparison of surface water and groundwater characteristics.
|
Characteristic
|
Surface Water
|
Ground Water
|
| Turbidity | high | low |
| Dissolved minerals | low-moderate | high |
| Biological content | high | low |
| Temporal variability | very high | low |
In contrast to surface supplies, groundwaters are relatively free from suspended contaminants, because they are filtered as they move through the strata. The filtration also removes most of the biological contamination. Some groundwaters with a high iron content contain sulfate reducing bacteria. These are a source of fouling and corrosion in industrial water systems.
Groundwater chemistry tends to be very stable over time. A groundwater may contain an undesirable level of scale forming solids, but due to its fairly consistent chemistry it may be treated effectively.
Mineral Reactions: As groundwater encounters different minerals, it dissolves them according to their solubility characteristics. In some cases chemical reactions occur, enhancing mineral solubility.
A good example is the reaction of groundwater with limestone. Water percolating from the surface contains atmospheric gases. One of these gases is carbon dioxide, which forms carbonic acid when dissolved in water. The decomposition of organic matter beneath the surface is another source of carbon dioxide. Limestone is a mixture of calcium and magnesium carbonate. The mineral, which is basic, is only slightly soluble in neutral water. The slightly acidic groundwater reacts with basic limestone in a neutralization reaction that forms a salt and a water of neutralization. The salt formed by the reaction is a mixture of calcium and magnesium bicarbonate. Both bicarbonates are quite soluble. This reaction is the source of the most common deposition and corrosion problems faced by industrial users. The calcium and magnesium (hardness) form scale on heat transfer surfaces if the groundwater is not treated before use in industrial cooling and boiler systems. In boiler feedwater applications, the thermal breakdown of the bicarbonate in the boiler leads to high levels of carbon dioxide in condensate return systems. This can cause severe system corrosion.
Structurally, limestone is porous. That is, it contains small holes and channels called "interstices". A large formation of limestone can hold vast quantities of groundwater in its structure. Limestone formations that contain these large quantities of water are called aquifers, a term derived from Latin roots meaning water bearing.
If a well is drilled into a limestone aquifer, the water can he withdrawn continuously for decades and used for domestic and industrial applications. Unfortunately, the water is very hard, due to the neutralization/dissolution reactions described above. This necessitates extensive water treatment for most uses.
CHEMICAL REACTIONS
Numerous chemical tests must be conducted to ensure effective control of a water treatment program. Most of these tests are addressed in detail in Chapters 39-71. Because of their significance in many systems, three tests, pH, alkalinity, and silica, are discussed here as well.
pH Control
Good pH control is essential for effective control of deposition and corrosion in many water systems. Therefore, it is important to have a good understanding of the meaning of pH and the factors that affect it.
Pure H2O exists as an equilibrium between the acid species, H+ (more correctly expressed as a protonated water molecule, the hydronium ion, H30+) and the hydroxyl radical, OH -. In neutral water the acid concentration equals the hydroxyl concentration and at room temperature they both are present at 10-7 gram equivalents (or moles) per liter.
The "p" function is used in chemistry to handle very small numbers. It is the negative logarithm of the number being expressed. Water that has 10-7 gram equivalents per liter of hydrogen ions is said to have a pH of 7. Thus, a neutral solution exhibits a pH of 7. Table 1-3 lists the concentration of H+ over 14 orders of magnitude. As it varies, the concentration of OH - must also vary, but in the opposite direction, such that the product of the two remains constant.
Table 1-3. pH relationships.
| pHa |
H+ Concentration Exponential Notation, gram moles/L
|
H+ Concentration, Normality
|
OH - Concentration, Normality
|
OH - Concentration, Exponential Notation, gram moles/L
|
pOH -
|
| 0 |
100
|
1 | 0.00000000000001 |
10-14
|
14 |
| 1 |
10-1
|
0.1 | 0.0000000000001 |
10--13
|
13 |
| 2 |
10-2
|
0.01 | 0.000000000001 |
10--12
|
12 |
| 3 |
10-3
|
0.001 | 0.00000000001 |
10-11
|
11 |
| 4 |
10-4
|
0.0001 | 0.0000000001 |
10-10
|
10 |
| 5 |
10-5
|
0.00001 | 0.000000001 |
10-9
|
9 |
| 6 |
10-6
|
0.000001 | 0.00000001 |
10-8
|
8 |
| 7 |
10-7
|
0.0000001 | 0.0000001 |
10-7
|
7 |
| 8 |
10-8
|
0.00000001 | 0.000001 |
10-6
|
6 |
| 9 |
10-9
|
0.000000001 | 0.00001 |
10-5
|
5 |
| 10 |
10-10
|
0.0000000001 | 0.0001 |
10-4
|
4 |
| 11 |
10-11
|
0.00000000001 | 0.001 |
10-3
|
3 |
| 12 |
10-12
|
0.000000000001 | 0.01 |
10-2
|
2 |
| 13 |
10-13
|
0.0000000000001 | 0.1 |
10-1
|
1 |
| 14 |
10-14
|
0.00000000000001 | 1 |
100
|
0 |
apH+pOH=14.
Confusion regarding pH arises from two sources:
- the inverse nature of the function
- the pH meter scale
It is important to remember that as the acid concentration increases, the pH value decreases (see Table 1-4).
Table 1-4. Comparative pH levels of common solutions.
| 12 | OH - alkalinity 500 ppm as CaCO3 |
| 11 | OH - alkalinity 50 ppm as CaCO3 Columbus. OH, drinking water, a |
| 10 | OH - alkalinity 5 ppm as CaCO3 |
| 9 | strong base anion exchanger effluents |
| 8 | phenolphthalein end point |
| 7 | neutral point at 25 °C |
| 6 | Weymouth, NIA, drinking water, a |
| 5 | methyl orange end point |
| 4 | FMA 4 ppm as CaCO3 |
| 3 | FMA 40 ppm as CaCO3 strong acid cation exchanger effluent |
| 2 | FMA 400 ppm as CaCO3 |
a Extremes of drinking water pH
The pH meter can be a source of confusion, because the pH scale on the meter is linear, extending from 0 to 14 in even increments. Because pH is a logarithmic function, a change of I pH unit corresponds to a 10 fold change in acid concentration. A decrease of 2 pH units represents a 100 fold change in acid concentration.
Alkalinity
Alkalinity tests are used to control lime-soda softening processes and boiler blowdown and to predict the potential for calcium scaling in cooling water systems. For most water systems, it is important to recognize the sources of alkalinity and maintain proper alkalinity control.
Carbon dioxide dissolves in water as a gas. The dissolved carbon dioxide reacts with solvent water molecules and forms carbonic acid according to the following reaction:
CO2 + H2O = H2CO3
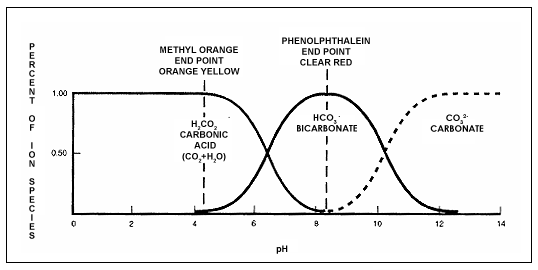
Only a trace amount of carbonic acid is formed, but it is acidic enough to lower pH from the neutral point of 7. Carbonic acid is a weak acid, so it does not lower pH below 4.3. However, this level is low enough to cause significant corrosion of system metals.
If the initial loading of CO2 is held constant and the pH is raised, a gradual transformation into the bicarbonate ion HCO3- occurs. This is shown in see Figure 1-2.
The transformation is complete at pH 8.3. Further elevation of the pH forces a second transformation into carbonate, CO32-. The three species carbonic acid, bicarbonate, and carbonate can be converted from one to another by means of changing the pH of the water.
Variations in pH can be reduced through "buffering" the addition of acid (or caustic). When acid (or caustic) is added to a water containing carbonate/bicarbonate species, the pH of the system does not change as quickly as it does in pure water. Much of the added acid (or caustic) is consumed as the carbonate/bicarbonate (or bicarbonate/carbonic acid) ratio is shifted.
Alkalinity is the ability of a natural water to neutralize acid (i.e., to reduce the pH depression expected from a strong acid by the buffering mechanism mentioned above). Confusion arises in that alkaline pH conditions exist at a pH above 7, whereas alkalinity in a natural water exists at a pH above 4.4.
Alkalinity is measured by a double titration; acid is added to a sample to the Phenolphthalein end point (pH 8.3) and the Methyl Orange end point (pH 4.4). Titration to the Phenolphthalein end point (the P-alkalinity) measures OH - and 1/2 CO32-; titration to the Methyl Orange end point (the M-alkalinity) measures OH -, CO32- and HCO3 .
Silica
When not properly controlled, silica forms highly insulating, difficult to remove deposits in cooling systems, boilers, and turbines. An understanding of some of the possible variations in silica testing is valuable.
Most salts, although present as complicated crystalline structures in the solid phase, assume fairly simple ionic forms in solution. Silica exhibits complicated structures even in solution.
Silica exists in a wide range of structures, from a simple silicate to a complicated polymeric material. The polymeric structure can persist when the material is dissolved in surface waters.
The size of the silica polymer can be substantial, ranging up to the colloidal state. Colloidal silica is rarely present in groundwaters. It is most commonly present in surface waters during periods of high runoff.
The polymeric form of silica does not produce color in the standard molybdate based colorimetric test for silica. This form of silica is termed "nonreactive". The polymeric form of silica is not thermally stable and when heated in a boiler reverts to the basic silicate monomer, which is reactive with molybdate.
As a result, molybdate testing of a boiler feedwater may reveal little or no silica, while boiler blowdown measurements show a level of silica that is above control limits. High boiler water silica and low feedwater values are often a first sign that colloidal silica is present in the makeup.
One method of identifying colloidal silica problems is the use of atomic emission or absorption to measure feedwater silica. This method, unlike the molybdate chemistry, measures total silica irrespective of the degree of polymerization.
Learn more about the various industries served by Veolia's various industrial water treatment applications.
Figure 1-1. Global water cycle.(Source: U.S. Geological Survey)
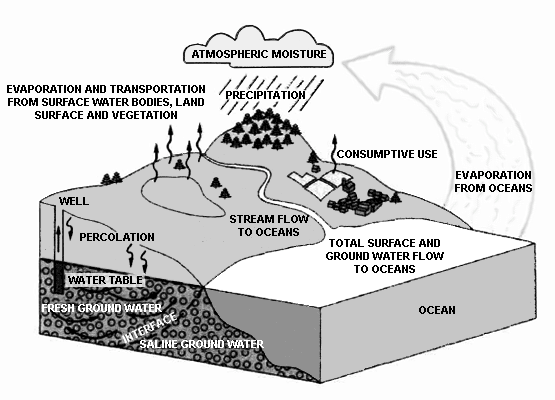
Figure 1-2. Carbonic acid, bicarbonate and carbonate distribution as a function of pH.