What is USP <645> and why does it matter?
USP <645> is a general chapter in the United States Pharmacopeia (USP) that provides procedures for measuring the conductivity of water. It establishes specific test methods and acceptance criteria for different types of pharmaceutical water including purified water (PW), water for injection (WFI), water for hemodialysis, and other sterile waters.
Electrical conductivity is a measure of a substance’s ability to transmit electric charge. In water quality applications, conductivity results from dissolved ionic material. In highly controlled pharmaceutical water systems, conductivity changes often indicate contamination, though conductivity also serves as a gauge of water composition, chemistry, and consistency. Because of the relative speed and ease of measuring conductivity, this parameter has long been regulated in many applications and is widely used as an early indicator of water quality changes.
Salts, dissolved carbon dioxide (CO2) and high pH levels increase ions in solution, thereby increasing conductivity. Fluctuations in this measurement indicate the presence and level of ionic impurities, providing data about contamination in the sample.
USP <645> works in tandem with USP <643> “Total Organic Carbon”, which details the monitoring of organic carbon present in a sample. Together, these essential chemical limit tests determine whether there is sufficient control in the water purification system. TOC and conductivity provide non-specific assessments of water quality, meaning they do not identify specific contaminant species, but rather measure entire classes of contaminants. This pair of tests quantifies contamination levels that could indicate microbial growth, residues from insufficient cleaning processes, or other impurities.
Conductivity, TOC, endotoxin and bioburden comprise four critical parameters that must be monitored to ensure PW and WFI meet compendial requirements for quality control (QC).
Download Now: Conductivity and TOC Sampling - Standard Operating Procedure (SOP) for Pharmaceutical Grade Ultrapure Water
Conductivity and TOC monitoring provide necessary information to confirm PW and WFI production is free of process by-products. For example, while pre-conditioning treatments such as ozone and electrodeionization (EDI) reduce contamination, they can themselves contribute contaminants if not properly managed. Conversely, while physical processes such as reverse osmosis and ultrafiltration do not introduce substances to the water, monitoring ensures these systems remain functional and effective.
Who is impacted by USP <645>?
USP <645> affects facilities who produce pharmaceutical-grade waters, use these waters as an ingredient, and instrument manufacturers.
Industries and sectors impacted by USP <645> include:
- Pharmaceutical and biopharmaceutical manufacturers
- Medical device manufacturers Ingredient manufacturers
- Research and development facilities and research institutions
- Contract testing and quality control laboratories
- Water purification system and instrument manufacturers
- Some healthcare and veterinary facilities
Additionally, Engineering Design Consultants (EDCs) and Engineering, Procurement, and Construction (EPC) contractors serving life science industries should take these regulations into consideration for point-of-use testing.
Note: This list is not exhaustive.
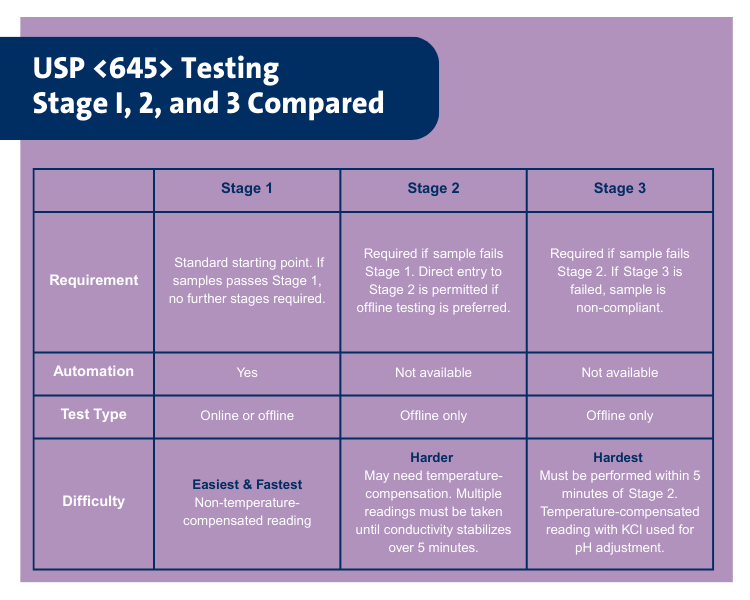
USP <645> overview and Stage I, II, and III testing
Testing methods detailed in USP chapter 645 follow a staged process of increasing complexity. They accommodate both online and offline testing scenarios with considerations for determining sample container suitability. It should be noted that Stage 1 is required for all samples, while Stages 2 and 3 are utilized only when a sample fails to meet Stage 1 test limits.
For pharmaceutical manufacturers, the most desirable state for compliance with USP <645> is Stage 1 conductivity testing. It is the simplest to execute, requiring the least time per sample. Automating Stage 1 USP <645> testing offers significant time savings as well as increased data integrity and security. Currently, there is no readily available automation solution for Stage 2 or Stage 3 testing.
Conductivity limits for Stage 1 range from 0.6 µS/cm at 0°C to 3.1 µS/cm at 100°C with specific conductivity requirements detailed in 5° increments. If the measured conductivity exceeds the table value, testing must proceed to Stage 2.
Stage 2 conductivity details testing procedures such as temperature adjustments, agitation, and observation in order to document the change in conductivity due to uptake of atmospheric carbon dioxide. If the reading is less than a net of 0.1 µS/cm per 5 minutes, the conductivity is noted. The conductivity limit for Stage 2 is 2.1 µS/cm. Conductivity measurements at this stage may be temperature-compensated to 25°C. If this limit is exceeded, testing must proceed to Stage 3.
Stage 3 is performed within five minutes of the Stage 2 conductivity determination. Sample temperature is maintained at 25±1°C with the addition of potassium chloride. pH is determined to the nearest 0.1 pH unit. The conductivity limit is determined at the measured pH value. pH-dependent conductivity limits range from 4.7 µS/cm at pH 5.0 to 4.6 µS/cm at pH 7.0, with conductivity requirements detailed in increments of 0.1 of pH.
Read More: Best Practices for Pharmaceutical Water Conductivity Testing
Download Now: Best Practice for Analyzing Compendia Water Samples for USP <643> and <645>
Download Now: Electrical Conductivity, Temperature Dependence and Instrument Methodology
USP <645> sampling methods
As mentioned above, USP <645> regulations accommodate both online and offline (laboratory) testing.
Online testing for USP <645>
Online testing involves measuring samples using an instrument with an integrated conductivity cell. Samples are automatically gathered and tested while data is recorded seamlessly, providing continuous monitoring. This applies to Stage 1 testing only. Stage 2 and Stage 3 testing are performed infrequently, and are not online tests.
Download Now: Conductivity and TOC Sampling Standard Operating Procedure (SOP)
Download Now: Low Level Linearity Conductivity Study
Offline or benchtop testing for USP <645>
Offline applications typically rely on portable meters or laboratory-based testing, where a quality control analyst gathers samples to perform periodic measurements. While compliant, the choice of analytical approach or instrument selection can significantly impact efficiency and data integrity.
For example, when conductivity measurements are made using a meter and probe, analysts sample and capture data manually, which increases the potential for transcription errors and reduces lab efficiency. These concerns can be addressed by using benchtop instruments that test for both conductivity and total organic carbon (TOC) simultaneously using the same sample vial. This approach offers significant efficiency gains, improved data integrity, and fewer opportunities for error.
USP <645> does not make specific recommendations for process development, testing location, testing frequency, or instrument selection. These choices should be based on suitability, manufacturing process, and intended use.
Download Now: Conductivity Bridge Study: From Benchtop Meter and Probe to Automated Analysis
USP <645> Conductivity validation and verification
The regulation is instrument-agnostic, but mandates cell constant verification for all methods. Additionally, the chapter does not explicitly specify verification frequency or concentration levels, but regular verification is compulsory to ensure compliance.
The cell constant must be known within ±2% accuracy and can be verified through two approaches:
- Directly, using solutions of known or traceable conductivity
- Indirectly, by comparing instrument readings with conductivity sensors having known or traceable cell constants
When necessary, cell constant adjustments should follow manufacturer protocols, with verification frequency determined by sensor design characteristics.
Several factors can compromise conductivity stability, with atmospheric CO2 being a primary concern. At low conductivity levels, samples are particularly susceptible to misreporting due to CO2 absorption and desorption effects, which can introduce unintended measurement bias. In contrast, higher-level samples are less immune to dissolved CO2 impacts but the compendial acceptance criteria of ±2% from stated values becomes more representative of actual instrument performance when using reference materials at elevated conductivity levels.
Resistance measurement calibration provides additional verification by replacing conductivity sensor electrodes with NIST-traceable precision resistors (accurate to ±0.1%). The measured conductivity with traceable resistors must fall within ±0.1 μS/cm of calculated values, requiring instruments to maintain minimum resolution of 0.1 μS/cm on the lowest range.
System verification ensures proper performance by comparing conductivity readings between the user's system and external calibrated devices. Values should be within ±5% of each other or meet acceptable differences based on water criticality and conductivity ranges, with sensors positioned to measure identical samples under the same temperature and water quality conditions.
Many companies that must comply with USP <645> go beyond basic compendial cell constant verification by implementing method suitability checks using different concentrations and acceptance criteria based on process capabilities. These additional checks are not regulatory requirements but provide facilities with additional confidence in instrument suitability for specific methods. These voluntary methods are separate from mandatory compendial verifications and should not be treated as a stand-in for compendial assessments.
Can you test for conductivity at the same time as TOC?
Conductivity and TOC can be tested simultaneously from the same sample vial, but typically they require different analytical methods and instruments. The tests are compatible and do not interfere with each other when performed on the same sample.
Performing simultaneous Stage 1 conductivity and TOC testing from the same vessel requires that the vessel not contribute in any significant way to either conductivity or TOC. Additionally, the vial must minimize ionic and organic contamination for compliance with USP <645> and USP <643>.
Download Now: Lean and Efficient Labs Need Simultaneous Testing for TOC and Conductivity
Download Now: Case Study: Improved Efficiency and Lower Costs Using Simultaneous Testing for TOC and Conductivity
Learn More: Standards and Vials
Download Now: Dual Use Conductivity and TOC (DUCT) Vials
We can support you with USP <645> compliance
Our experts can provide guidance on selecting appropriate instrumentation, applying proper testing procedures, and establishing effective SOPs for documentation and compliance.
After selecting suitable and compliant technology, comprehensive instrument qualification and method validation must be completed before data can be used for making quality decisions. Our industry-leading expertise ensures you are supported with thorough method validation and deployment strategies. With decades of specialized experience, we have refined these processes to meet the most stringent regulatory requirements while maintaining operational efficiency.
Modern efficiency improvements now enable dual testing of conductivity and TOC for compliance with USP <645> and USP <643> from a single sample using specialized vials that prevent ionic leaching and CO2 contamination. This approach enhances sample integrity while reducing analysis time, when compared to traditional methods.
Additionally, many facilities have adopted online water monitoring for real-time release testing (RTRT), eliminating the need for manual sampling and streamlining the QC process. This implementation of process analytical technology (PAT) provides immediate measurement of quality attributes and demonstrates process control in a validated state, particularly when using compatible membrane conductometric technology.
Download Now: Sievers M9: Ensuring Compliance with EP Water Monographs, EP 2.2.38, and USP <645> Conductivity Regulations
Download Now: Sievers M9 Analyzers Offer Simultaneous TOC and Conductivity Compendia Compliance Testing
Download Now: Low-Level Third-Party Conductivity Standards
- Tony Saavedra, MBA
-
Tony Saavedra, MBA is the Life Sciences Product Manager for Veolia’s Sievers Analytical Instruments product line, focusing on Sievers total organic carbon (TOC) software and instrumentation. Tony began his tenure with the Sievers product line as part of GE Analytical Instruments in 2011 in the field service organization and later held responsibility for leading the North America Technical Services team where he oversaw technical support, factory service, and refurbishment process operations. Prior to his positions with Sievers products, Tony served for 10 years in the US Navy where he supervised flight inspections and performed quality assurance inspections and maintenance of complex electronic radar and communication suites. Tony holds a BS in Electronic Engineering Technology from ECPI University and an MBA from Colorado State University.
- Sydney Jannetta
-
Sydney Jannetta is a Marketing Manager at Veolia, focusing on Sievers Instruments. Sydney has supported Sievers instrument customers for the last six years with expertise in TOC and endotoxin applications. She has provided method development services and feasibility testing to pharmaceutical manufacturers and has presented at over 20 national conferences. Sydney holds a Bachelor of Science degree in Chemistry from the University of Northern Colorado..
