- Pollutants
- Removal of insoluble contaminants
- Removal of soluble contaminants
- Solid waste handling
- Environmental regulations
Many industries use large volumes of water in their manufacturing operations. Because some of this water becomes contaminated, it requires treatment before discharge (Figure 37-1).
Improvements in determining the effects of industrial waste discharges have led to the adoption of stringent environmental laws, which define the degree of treatment necessary to protect water quality. Discharge permits, issued under the National Pollutant Discharge Elimination System (NPDES), regulate the amount of pollutants that an industry can return to the water source. The permitted quantities are designed to ensure that other users of the water will have a source that meets their needs, whether these needs are for municipal water supply, industrial or agricultural uses, or fishing and recreation. Consideration is given to the feasibility of removing a pollutant, as well as the natural assimilative capacity of the receiving stream. This assimilative capacity varies with the type and amount of pollutant.
Wastewater treatment plants are designed to convert liquid wastes into an acceptable final effluent and to dispose of solids removed or generated during the process. In most cases, treatment is required for both suspended and dissolved contaminants. Special processes are required for the removal of certain pollutants, such as phosphorus or heavy metals.
Wastewater can be recycled for reuse in plant processes to reduce disposal requirements (Figure 37-2). This practice also reduces water consumption.
Organic Compounds
The amount of organic material that can be discharged safely is defined by the effect of the material on the dissolved oxygen level in the water. Organisms in the water use the organic matter as a food source. In a biochemical reaction, dissolved oxygen is consumed as the end products of water and carbon dioxide are formed. Atmospheric oxygen can replenish the dissolved oxygen supply, but only at a slow rate. When the organic load causes oxygen consumption to exceed this resupply, the dissolved oxygen level drops, leading to the death of fish and other aquatic life. Under extreme conditions, when the dissolved oxygen concentration reaches zero, the water may turn black and produce foul odors, such as the "rotten egg" smell of hydrogen sulfide. Organic compounds are normally measured as chemical oxygen demand (COD) or biochemical oxygen demand (BOD).
Nutrients
Nitrogen and phosphorus are essential to the growth of plants and other organisms. However, nitrogen compounds can have the same effect on a water source as carbon-containing organic compounds. Certain organisms use nitrogen as a food source and consume oxygen.
Phosphorus is a concern because of algae blooms that occur in surface waters due to its presence. During the day, algae produce oxygen through photosynthesis, but at night they consume oxygen.
Solids
Solids discharged with a waste stream may settle immediately at the discharge point or may remain suspended in the water. Settled solids cover the bottom-dwelling organisms, causing disruptions in population and building a reservoir of oxygen-consuming materials. Suspended solids increase the turbidity of the water, thereby inhibiting light transmittance. Deprived of a light source, photosynthetic organisms die. Some solids can coat fish gills and cause suffocation.
Acids and Alkalies
The natural buffering system of a water source is exhausted by the discharge of acids and alkalies. Aquatic life is affected by the wide swings in pH as well as the destruction of bicarbonate alkalinity levels.
Metals
Certain metals are toxic and affect industrial, agricultural, and municipal users of the water source. Metals can cause product quality problems for industrial users. Large quantities of discharged salts necessitate expensive removal by downstream industries using the receiving stream for boiler makeup water.
REMOVAL OF INSOLUBLE CONTAMINANTS
Various physical methods may be used for the removal of wastewater contaminants that are insoluble in water, such as suspended solids, oil, and grease. Ordinarily, water-soluble contaminants are chemically converted to an insoluble form to allow removal by physical methods. Essentially, biological waste treatment is this conversion of soluble contaminants to insoluble forms.
Gravity Separation
Most waste treatment systems employ a gravity separation step for suspended particle or oil removal.
The settling rate of a particle is defined in terms of "free" versus "hindered" settling. A free settling particle's motion is not affected by that of other particles, the vessel's walls, or turbulent currents. A particle has a hindered settling rate if there is any interference from these effects.
The free settling of a discrete particle in a rising fluid can be described as the resolution of several forces-gravity, the drag exerted on the particle, and the buoyant force as described by Archimedes' principle. The particle's velocity increases until it reaches a terminal velocity as determined by these forces. The terminal velocity is then:
where:
v = velocity, ft/sec
g = gravitation constant, ft/sec2
mP = mass of the particle, lb
rP = density of the particle, lb/ft3
rf = density of the fluid, lb/ft3
Aa = cross sectional area of the particle ex-posed to the direction of motion, ft2
Cd = drag coefficient, a function of particle geometry
Gravity settling is employed primarily for removal of inorganic suspended solids, such as grit and sand. Therefore, in the approximation of the drag coefficient, it is assumed that particles are spherical. Further, if a Reynolds number of less than 2.0 is assumed, the settling velocity of a discrete particle can be described by Stokes' settling equation:
| V = |
GdP2(rP - rf) |
| 18µ |
where:
DP = particle diameter, ft
µ = fluid viscosity, lb/ft-sec
The terminal velocity of a particle in the "free" settling zone is a function of its diameter, the density difference between the particle and the fluid, and the fluid viscosity.
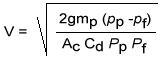
The equipment employed for gravity separation for waste treatment is normally either a rectangular basin with moving bottom scrapers for solids removal or a circular tank with a rotating bottom scraper. Rectangular tanks are normally sized to decrease horizontal fluid velocity to approximately 1 ft/min. Their lengths are three to five times their width, and their depths are 3-8 ft.
Circular clarifiers (see Figure 37-3) are ordinarily sized according to surface area, because velocity must be reduced below the design particle's terminal velocity. The typical design provides a rise rate of 600-800 gpd/ft2.
When wastewater contains appreciable amounts of hydrocarbons, removal of these contaminants becomes a problem. Oil is commonly lower in density than water; therefore, if it is not emulsified, it can be floated in a separate removal stage or in a dual-purpose vessel that allows sedimentation of solids. For example, the refining industry uses a rectangular clarifier with a surface skimmer for oil and a bottom rake for solids as standard equipment. This design, specified by the American Petroleum Institute, is designated as an API separator. The basic principles govern-ing the separation of oil from water by gravity differential are also expressed by Stokes' Law.
Air Flotation
Where the density differential is not sufficient to separate oil and oil-wetted solids, air flotation may be used to enhance oil removal. In this method, air bubbles are attached to the contaminant particles, and thus the apparent density difference between the particles is increased.
Dissolved air flotation (DAF) is a method of introducing air to a side stream or recycle stream at elevated pressures in order to create a supersaturated stream. When this stream is introduced into the waste stream, the pressure is reduced to atmospheric, and the air is released as small bubbles. These bubbles attach to contaminants in the waste, decreasing their effective density and aiding in their separation.
The most important operational parameters for contaminant removal by dissolved air flotation are:
- air pressure
- recycle or slip stream flow rate
- influent total suspended solids (TSS) including oil and grease
- bubble size
- dispersion
Air pressure, recycle, and influent TSS are normally related in an air-to-solids (A/S) ratio expressed as:
| A/S = | KSa(fP-1)R |
| SSQ |
where:
K = a constant, approximately 1.3
Sa = the solubility of air at standard conditions, mL/L
f = air dissolved/Sa, usually 0.5-0.8
P = operating pressure, atm
R = recycle rate, gpm
SS = influent suspended solids, mg/L
Q = wastewater flow, gpm
The A/S ratio is most important in determining effluent TSS. Recycle flow and pressure can be varied to maintain an optimal A/S ratio. Typical values are 0.02-0.06.
In a DAF system, the supersaturated stream may be the entire influent, a slip stream, fresh water, or a recycle stream. Recycle streams are most common, because pressurization of a high- solids stream through a pump stabilizes and disperses oil and oil-wetted solids.
As in gravity settling, air flotation units are designed for a surface loading rate that is a function of the waste flow and rise velocity of the contaminants floated by air bubbles. The retention time is a function of tank depth.
DAF units can be rectangular in design but are usually circular, resembling a primary clarifier or thickener. They are often single-stage units.
Induced air flotation (IAF) is another method of decreasing particle density by attaching air bubbles to the particles; however, the method of generating the air bubbles differs. A mechanical action is employed to create the air bubbles and their contact with the waste contaminants. The most common methods use high-speed agitators or recycle a slip stream through venturi nozzles to entrain air into the wastewater.
In contrast to DAF units, IAF units are usually rectangular and incorporate four or more air flotation stages in series. The retention time per stage is significantly less than in DAF circular tanks.
As in gravity settling, the diameter of the particle plays an important role in separation. Polyelectrolytes may be used to increase effective particle diameters. Polymers are also used to destabilize oil-water emulsions, thereby allowing the free oil to be separated from the water. Polymers do this by charge neutralization, which destabilizes an oil globule surface and allows it to contact other oil globules and air bubbles. Emulsion breakers, surfactants, or surface-active agents are also used in air flotation to destabilize emulsions and increase the effectiveness of the air bubbles.
Filtration
Filtration is employed in waste treatment wherever suspended solids must be removed. In practice, filtration is most often used to polish wastewater following treatment. In primary waste treatment, filters are often employed to remove oil and suspended solids prior to biological treatment. More commonly, filters are used following biological treatment, prior to final discharge or reuse.
Filters used for waste treatment may be designed with single-, dual-, or multimedia and may be of the pressure or gravity type.
REMOVAL OF SOLUBLE CONTAMINANTS
pH Adjustment-Chemical Precipitation
Often, industrial wastewaters contain high concentrations of metals, many of which are soluble at a low pH.
Adjustment of pH precipitates these metals as metal oxides or metal hydroxides. The pH must be carefully controlled to minimize the solubility of the contaminant. As shown in Figure 37-4, some compounds, such as zinc, are amphoteric and redissolve at a high pH. Chemicals used for pH adjustment include lime, sodium hydroxide, and soda ash.
Chemical precipitation of soluble ions often occurs as the result of pH adjustment. Contaminants are removed either by chemical reaction leading to precipitation or by adsorption of ions on an already formed precipitate.
Biological Oxidation-Biochemical Reactions
One of the most common ways to convert soluble organic matter to insoluble matter is through biological oxidation. Soluble organics metabolized by bacteria are converted to carbon dioxide and bacterial floc, which can be settled from solution.
Various microorganisms feed on dissolved and suspended organic compounds. This natural biodegradation can occur in streams and lakes. If the assimilative capacity of the stream is surpassed, the reduced oxygen content can cause asphyxiation of fish and other higher life forms. This natural ability of microorganisms to break down complex organics can be harnessed to remove materials within the confines of the waste plant, making wastewater safe for discharge.
The biodegradable contaminants in water are usually measured in terms of biochemical oxygen demand (BOD). BOD is actually a measure of the oxygen consumed by microorganisms as they assimilate organics.
Bacteria metabolize oxygen along with certain nutrients and trace metals to form cellular matter, energy, carbon dioxide, water, and more bacteria. This process may be represented in the form of a chemical reaction:
|
Food (organic compounds) + Microorganisms + Oxygen + Nutrients |
» |
Cellular matter + Microorganisms + Carbon dioxide + Water + Energy |
The purity of the water depends on minimizing the amount of "food" (organic compounds) that remains after treatment. Therefore, biological waste treatment facilities are operated to provide an environment that will maximize the health and metabolism of microorganisms. An integral part of the biological process is the conversion of soluble organic material into insoluble materials for subsequent removal (Figure 37-5). An overview of factors involved in biological oxidation is given in Table 37-1.
Open Lagoon Biological Oxidation
Where organic loads are low and sufficient land area is available, open lagoons may be used for biological treatment. Lagoons provide an ideal habitat for microorganisms. Natural infiltration of oxygen is sufficient for biological oxidation if the organic loading is not too high.However, mechanical aeration (Figure 37-6) is often used to increase the ability to handle a higher loading.
Lagoons are nothing more than long-term retention basins. Ordinarily shallow in depth, they depend on surface area, wind, and wave action for oxygen transfer from the atmosphere. Depending on the influent BOD loading and oxygen transfer, lagoons may be aerobic or anaerobic. Lagoons are used primarily for low BOD wastes or as polishing units after other biological operations.
Aerated Lagoons. As BOD loading increases, naturally occurring surface oxygen transfer becomes insufficient to sustain aerobic bacteria. It then becomes necessary to control the environment artificially by supplying supplemental oxygen. Oxygen, as air, is introduced either by mechanical agitators or by blowers and subsurface aerators. Because energy must be expended, the efficiency of the oxygen transfer is a consideration. Therefore, although unaerated lagoons are typically 3-5 ft deep, allowing large surface areas for natural transfer, aerated lagoons are usually 10-15 ft deep in order to provide a longer, more difficult path for oxygen to escape unconsumed. Aerated lagoons also operate with higher dissolved oxygen content.
Facultative Lagoons. Lagoons without mechanical aeration are usually populated by facultative organisms. These organisms have the ability to survive with or without oxygen. A lagoon designed specifically to be facultative is slightly deeper than an unaerated lagoon. Influent suspended solids and solids created by the metabolism of the aerobic bacteria settle to the bottom of the lagoon where they undergo further decomposition in an anaerobic environment.
Activated Sludge Oxidation
According to the reaction presented previously, control of contaminant oxidation at high BOD loadings requires a bacteria population that is equal to the level of food. This need is the basis for the activated sludge process.
In the activated sludge process, reactants, food, and microorganisms are mixed in a controlled environment to optimize BOD removal. The process incorporates the return of concentrated microorganisms to the influent waste.
When bacteria are separated from wastewater leaving an aeration basin and reintroduced to the influent, they continue to thrive. The recirculated bacteria continue to oxidize wastewater contaminants, and if present in sufficient quantity, produce a relatively low BOD effluent water.
Because the activated sludge process incorporates the return of concentrated microorganisms, it must include a process for microorganism concentration and removal. This process includes an aeration stage and a sedimentation stage (Figure 37-7). Because suspended solids are considered wastewater contaminants, the sedimentation stage accomplishes two functions: concentration of bacteria and removal of solids.
The operating parameters that affect the performance of any activated sludge process are BOD, microorganisms, dissolved oxygen, retention time, nutrient concentration, and the external influences of temperature and pH. In order to understand the various activated sludge designs, it is necessary to examine the relationship between available food and bacteria population.
If a seed culture of bacteria is introduced into a fixed amount of food, the conditions shown in Figure 37-8 are created. Initially, excess food is present; therefore, the bacteria reproduce in a geometric fashion. This is termed the "log growth phase." As the population increases and food decreases, a plateau is reached in population. From the inflection point on the curve to the plateau, population is increasing but at a decreasing rate. This is called the "declining growth phase." Once the plateau is crossed, the bacteria are actively competing for the remaining food. The bacteria begin to metabolize stored materials, and the population decreases. This area of the curve is termed "endogenous respiration." Eventually, the bacteria population and BOD are at a minimum.
Because activated sludge is a continuous, steady-state process, each plant operates at some specific point on this curve, as determined by the oxidation time provided. The point of operation determines the remaining bacteria population and BOD effluent.
Optimization of an activated sludge plant requires the integration of mechanical, operational, and chemical approaches for the most practical overall program. Mechanical problems can include excessive hydraulic loading, insufficient aeration, and short-circuiting. Operational problems may include spills and shock loads, pH shocks, failure to maintain correct mixed liquor concentration, and excessive sludge retention in the clarifier.
Various chemical treatment programs are described below. Table 37-2 presents a comparison of various treatment schemes.
Sedimentation. Because activated sludge depends on microorganism recirculation, sedimentation is a key stage. The settleability of the biomass is a crucial factor. As bacteria multiply and generate colonies, they excrete natural biopolymers. These polymers and the slime layer that encapsulates the bacteria influence the flocculation and settling characteristics of bacteria colonies. It has been determined empirically that the natural settleability of bacteria colonies is also a function of their position on the time chart represented in Figure 37-8. Newly formed colonies in the log growth phase are relatively non-settleable. At the end of the declining growth phase and the first part of the endogenous phase, natural flocculation is at an optimum. As the endogenous phase continues, colonies break up and floc particles are dispersed, decreasing the biomass settleability.
Although microbes are eventually able to break down most complex organics and can tolerate very poor environments, they are very intolerant of sudden changes in pH, dissolved oxygen, and the organic compounds that normally upset an activated sludge system. These upsets normally result in poor BOD removal and excessive carryover of suspended solids (unsettled microorganisms) in the final effluent.
Aeration. Aeration is a critical stage in the activated sludge process. Several methods of aeration are used:
High Rate Aeration. High rate aeration operates in the log growth phase. Excess food is provided, by recirculation, to the biomass population. Therefore, the effluent from this design contains appreciable levels of BOD (i.e., the oxidation process is not carried to completion). Further, the settling characteristics of the biomass produced are poor. High sludge return rates are necessary to offset poor settling and to maintain the relatively high biomass population. Poor settling increases the suspended solids content of the effluent. The relatively poor effluent produced limits this design to facilities which need only pretreatment before discharge to a municipal system. The advantage of high rate aeration is low capital investment (i.e., smaller tanks and basins due to the short oxidation time).
Conventional Aeration. The most common activated sludge design used by municipalities and industry operates in the endogenous phase, in order to produce an acceptable effluent in BOD and TSS levels. Conventional aeration represents a "middle of the road" approach because its capital and operating costs are higher than those of the high rate process, but lower than those of the extended aeration plants. As shown in Figure 37-8, the conventional plant operates in the area of the BOD curve where further oxidation time produces little reduction in BOD. Natural flocculation is optimum, so the required sedimentation time for removal of suspended solids from the effluent is minimized.
Extended Aeration. Extended aeration plants operate in the endogenous phase, but use longer periods of oxidation to reduce effluent BOD levels. This necessitates higher capital and operating costs (i.e., larger basins and more air). In conjunction with lower BOD, extended aeration produces a relatively high suspended solids effluent when optimum natural settling ranges are exceeded.
Extended aeration designs may be necessary to meet effluent BOD requirements when the influent is relatively concentrated in BOD or the wastes are difficult to biodegrade. Because extended aeration operates on the declining side of the biomass population curve, net production of excess solids is minimized, as shown in Table 37-3. Therefore, savings in sludge handling and disposal costs may offset the higher plant capital and operating costs required for extended aeration.
Step Aeration/Tapered Aeration. In a plug flow basin, the head of the basin receives the waste in its most concentrated form. Therefore, metabolism and oxygen demand are greatest at that point. As the waste proceeds through the basin, the rate of oxygen uptake (respiration rate) decreases, reflecting the advanced stage of oxidation.
Tapered aeration and step aeration reduce this inherent disadvantage. Tapered aeration provides more oxygen at the head of the basin and slowly reduces oxygen supply to match demand as the waste flows through the basin. This results in better control of the oxidation process and reduced air costs.
Step aeration modifies the introduction of influent waste. The basin is divided into several stages, and raw influent is introduced to each stage proportionately. All return microorganisms (sludge) are introduced at the head of the basin. This design reduces aeration time to 3-5 hr, while BOD removal efficiency is maintained. The shorter aeration time reduces capital expenses because a smaller basin can be used. Operating costs are similar to those of a conventional plant.
Contact Stabilization. Due to the highly efficient sorptive capabilities of activated biomass, the time necessary for the biomass to "capture" the colloidal and soluble BOD is approximately 30 min to 1 hr. Oxidation of fresh food requires the normal aeration time of 4-8 hr. In the contact stabilization design, relatively quick sorption time reduces aeration tank volume requirements. The influent waste is mixed with return biomass in the initial aeration tank (or contact tank) for 30-90 min. The entire flow goes to sedimentation, where the biomass and its captured organics are separated and returned to a reaeration tank. In the reaeration tank the wastes undergo metabolism at a high biomass population. The system is designed to reduce tank volume by containing the large majority of flow for a short period of time.
This process is not generally as efficient in BOD removal as the conventional plant process, due to mixing limitations in the contact basin. Operating costs are equivalent. Due to the unstabilized state of the biomass at sedimentation, flocculation is inferior. Suspended solids in the effluent are problematic.
Because this design exposes only a portion of the active biomass to the raw effluent at a time, it is less susceptible to feed variations and toxicants. For this reason it can be beneficial for treatment of industrial wastes.
Pure Oxygen Sludge Processes. Oxygen supply and transfer often become limiting factors in industrial waste treatment. As the name implies, pure oxygen activated sludge processes supply oxygen (90-99% O2) to the biomass instead of air. The increased partial pressure increases transfer rates and provides several advantages. Comparable or higher BOD removal efficiencies are maintained at higher BOD influent loadings and shorter retention times. Generally, aeration time is 2-3 hr. A further advantage is the production of lower net solids per pound of BOD removed. Therefore, sludge disposal costs are reduced.
The units are usually enclosed. Normally, three or four concrete box stages in series are provided for aeration. The raw wastewater, return biomass, and pure oxygen enter the first stage. Wastewater passes from stage to stage in the underflow.
The atmosphere flows over the open surface of each stage to the last stage, from which it is vented to control the oxygen content. Oxygen purity and the demand for oxygen decline through the stages. Each stage contains a mechanical agitator for mixing and oxygen transfer. By design, each stage is completely mixed. After aeration, the waste flows to a conventional sedimentation stage. BOD and TSS removals are usually somewhat better than in a conventional aeration system.
Chemical Treatment Programs. The following additives represent a variety of chemical programs that may be used to address problems and improve system efficiency.
Essential Nutrients. Nutrients, particularly nitrogen and phosphorus, may be added to ensure complete digestion of organic contaminants.
Polymers. Polymer feeding improves the settling of suspended solids. Cationic polymers can increase the settling rate of bacterial floc and improve capture of dispersed floc and cell fragments. This more rapid concentration of solids minimizes the volume of recycle flow so that the oxygen content of the sludge is not depleted. Further, the wasted sludge is usually more concentrated and requires less treatment for eventual dewatering. Polymers may also be used on a temporary basis to improve the removal of undesirable organisms, such as filamentous bacteria or yeast infestations, that cause sludge bulking or carryover of floating clumps of sludge.
Oxidizing Agents. Peroxide, chlorine, or other agents may be used for the selective oxidation of troublesome filamentous bacteria.Antifoam Agents. Antifoam agents may be used to control excessive foam.
Coagulants. In addition to antifoam agents, coagulants may be fed continuously to improve efficiency, or to address particularly difficult conditions. They may also be used intermittently to compensate for hydraulic peak loads or upset conditions.
Fixed Media Biological Oxidation
In contrast to activated sludge, in which the bio-mass is in a fluid state, fixed media oxidation passes influent wastewater across a substructure laden with fixed biomass. The parameters for healthy microorganisms remain the same, except the manner in which food and microorganisms come into contact.
Fixed media designs allow a biological slime layer to grow on a substructure continually exposed to raw wastewater. As the slime layer grows in thickness, oxygen transfer to the innermost layers is impeded. Therefore, mixed media designs develop aerobic, facultative, and anaerobic bacteria as a function of the thickness of the slime layer. Eventually, either because of size and wastewater shear or the death of the microorganisms, some of the slime layer sloughs off. In a continuous process, this constantly sloughing material is carried to a sedimentation stage, where it is removed. There are no provisions to recycle the microorganisms, because return sludge would plug the fixed media structure. In fact, media plugging and lack of oxygen transfer are the primary difficulties encountered with fixed media designs. Plugging problems can be alleviated by increased wastewater shear. This is normally accomplished by recycling of a portion of the effluent wastewater.
Trickling Filters. Trickling filters are not really filters but a filter-like form of fixed media oxidation. Wastewater is sprayed over a bed of stones, 3-5 in. in diameter. Bed depths range from 5 to 7 ft. Because air contact is the sole means of oxygen transfer, microorganisms become more oxygen deficient as depth increases.
Trickling filters can be classified by hydraulic loading as low-rate, high-rate, or roughing. Due to inherent oxygen transfer difficulties, even low rate filters cannot achieve the BOD removal possible in conventional activated sludge systems. Industrial trickling filters are usually followed by an activated sludge unit. They may be used as a pretreatment step before discharge to a municipal sewage system.
Biological Towers. Another form of fixed media filter uses synthetic materials in grid fashion as a substructure for biological growth. The high porosity available with artificially designed media alleviates the oxygen transfer problems of trickling filters and allows greater bed depths. Bed depths of up to 20 ft with adequate oxygen allow longer contact and consequently better BOD removal.
Biodiscs. Biodiscs are a recently developed form of fixed media oxidation. The media is fixed to a rotating shaft that exposes the media alternately to food (wastewater) and oxygen (atmosphere). Design parameters include speed of rotation, depth of the wastewater pool, porosity of the synthetic media, and number of series and parallel stages. These units circumvent the oxygen limitations of the trickling filter and therefore provide BOD removal comparable to conventional activated sludge systems. Solids produced are easily settled in the sedimentation stage, provi-ding acceptable TSS levels in the effluent. Little operational attention is required.
Wastewater treatment is a concentration process in which waterborne contaminants are removed from the larger wastewater stream and concentrated in a smaller side stream. The side stream is too large to be disposed of directly, so further concentration processes are required. These processes are called "solid waste handling" operations.
Stabilization/Digestion
Sludge stabilization is a treatment technique applied to biological sludge to reduce its odor-causing or toxic properties. This treatment often reduces the amount of solids as a side effect. Anaerobic and aerobic digestion, lime treatment, chlorine oxidation, heat treatment, and composting fall into this category.
Anaerobic Digestion. Anaerobic digestion takes place in an enclosed tank, as depicted in Figure 37-9. The biochemical reactions take place in the following phases:
| Organics | + | Acid-forming organisms | » |
Volatile acids |
||
| Volatile acids | + | Methane formers | » | Methane | + |
Carbon dioxide |
Sludge solids are decreased due to the conversion of biomass to methane and carbon dioxide. The methane can be recovered for its heating value.
Aerobic Digestion. Aerobic digestion is the separate aeration of sludge in an open tank. Oxidation of biodegradable matter, including cell mass, occurs under these conditions. As in anaerobic digestion, there is a decrease in sludge solids, and the sludge is well stabilized with respect to odor formation. Capital costs are lower than those of anaerobic digestion, but operating costs are higher, and there is no by-product methane production.
Lime Treatment. Stabilization by lime treatment does not result in a reduction of organic matter. Addition of sufficient lime to maintain the pH of the sludge above 11.0 for 1-14 days is considered sufficient to destroy most bacteria.
Composting. A natural digestion process, composting usually incorporates sludge material that later will be applied to farmland. Sludge is combined with a bulking material, such as other solid wastes or wood chips, and piled in windrows. Aeration is provided by periodic turning of the sludge mass or by mechanical aerators. The energy produced by the decomposition reaction can bring the waste temperature to 140-160°F, destroying pathogenic bacteria. At the end of the composting period, the bulking material is separated, and the stabilized sludge is applied to land or sent to a landfill.
Sludge Conditioning
Typically, sludge from a final liquid-solids separation unit may contain from 1 to 5% total suspended solids. Figure 37-10 shows the relationship between the volume of sludge to be handled and the solids content in the sludge. Because of the cost savings associated with handling smaller volumes of sludge, there is an economic incentive to remove additional water. Dewatering equipment is designed to remove water in a much shorter time span than nature would by gravity. Usually, an energy gradient is used to promote rapid drainage. This requires frequent conditioning of the sludge prior to the dewatering step.
Conditioning is necessary due to the nature of the sludge particles. Both inorganic and organic sludge consist of colloidal (less than 1 µm), intermediate, and large particles (greater than 200 µm). The large particles, or flocs, are usually compressible. Under an energy gradient, these large flocs compress and prevent water from escaping. The small particles also participate in this mechanism, plugging the pores of the sludge cake, as shown in Figure 37-11. The pressure drop through the sludge cake, due to the decrease in porosity and pore sizing, exceeds available energy, and dewatering ceases.
The purpose of sludge conditioning is to provide a rigid sludge structure of a porosity and pore size sufficient to allow drainage. Biological sludges are conditioned with FeCl3, lime, and synthetic cationic polymers, either separately or in combination. Heat conditioning and low-pressure oxidation are also used for biological sludges. Inorganic sludges are conditioned with FeCl3, lime, and either cationic or anionic polymers.
Dewatering
Belt Filter Press. Belt filter presses have been used in Europe since the 1960's and in the United States since the early 1970's. They were initially designed to dewater paper pulp and were subsequently modified to dewater sewage sludge.
Belt filter presses are designed on the basis of a very simple concept. Sludge is sandwiched between two tensioned porous belts and passed over and under rollers of various diameters. At a constant belt tension, rollers of decreasing diameters exert increasing pressure on the sludge, thus squeezing out water. Although many different designs of belt filter presses are available, they all incorporate a polymer conditioning unit, a gravity drainage zone, a compression (low-pressure) zone, and a shear (high-pressure) zone. Figure 37-12 shows these zones in a simplified schematic of a belt filter press.
Polymer Conditioning Unit. Polymer conditioning can take place in a small tank, in a rotating drum attached to the top of the press, or in the sludge line. Usually, the press manufacturer supplies a polymer conditioning unit with the belt filter press.
Gravity Drainage Zone. The gravity drainage zone is a flat or slightly inclined belt, which is unique to each press model. In this section, sludge is dewatered by the gravity drainage of free water. The gravity drainage zone should increase the solids concentration of the sludge by 5-10%. If the sludge does not drain well in this zone, the sludge can squeeze out from between the belts or the belt mesh can become blinded. The effectiveness of the gravity drainage zone is a function of sludge type, quality, and conditioning, along with the screen mesh and the design of the drainage zone.
Compression (Low-Pressure) Area. The compression, or low-pressure, area is the point at which the sludge is "sandwiched" between the upper and lower belts. A firm sludge cake is formed in this zone in preparation for the shear forces encountered in the high-pressure zone.
Shear (High-Pressure) Zone. In the shear, or high-pressure, zone, forces are exerted on the sludge by the movement of the upper and lower belts, relative to each other, as they go over and under a series of rollers with decreasing diameters. Some manufacturers have an independent high-pressure zone which uses belts or hydraulic cylinders to increase the pressure on the sludge, producing a drier cake. A dry cake is particularly important for plants that use incineration as the final disposal.
Dewatering belts are usually woven from monofilament polyester fibers. Various weave combinations, air permeabilities, and particle retention capabilities are available. These parameters greatly influence press performance.
Usually, cationic polymers are used for sludge conditioning. A two-polymer system is often used on a belt filter press to improve cake release from the upper dewatering belt. The polymer must be selected carefully to ensure optimum performance.
Odors are controlled by proper ventilation, by ensuring that the sludge does not turn septic, and by the use of added chemicals, such as potassium permanganate or ferric sulfate, to neutralize the odor-causing chemicals.
Screw Press. A recent development in sludge dewatering equipment, used primarily in the pulp and paper industry, is the screw press. Screw presses are most effective for primary sludges, producing cake solids of 50-55%, but are also appropriate for primary and secondary blended sludges.
Sludge is conditioned and thickened prior to dewatering. The conditioned sludge enters one end of the machine, as shown in Figure 37-13. A slowly rotating screw, analogous to a solid bowl centrifuge, conveys and compresses the solids.
The screw has the same outer diameter and pitch for the entire length of the press. In some models, the diameter of the screw shaft increases toward the discharge end of the screw press to enhance dewatering. The compression ratio (the ratio of free space at the inlet to the space at the discharge end of the screw) is selected according to the nature of the material to be dewatered and the dewatering requirement. Dewatered cake is discharged as it is pressed against the spring or hydraulically loaded cone mounted at the end of the screw press.
The drum of the screw press consists of a fine strainer screen, a thicker punched holding plate, and a reinforcement rib.
Filtrate is collected in the collecting pan located under the screw press, and the cake is transported to the next stage.
Vacuum Filters. Vacuum filtration uses various porous materials as filter media, including cloth, steel mesh, and tightly wound coil springs. Under an applied vacuum, the porous medium retains the solids, but allows water to pass through. The relative importance of cake dryness, filtrate quality, and filter cake yield can vary from one system to another.
A decrease in drum speed allows more time for drying of the sludge to increase cake dryness. However, this also decreases the filter cake yield, defined as pounds of dry solids per hour per square foot of filter area. Polymers can help produce a drier cake without the problem of a lower filter cake yield. Synthetic polymers improve cake dryness by agglomerating sludge particles that may hinder the removal of water. This agglomeration also increases the solids capture across the unit, which results in a higher-quality filtrate.
Centrifuges. Centrifugal force, 3500-6000 times the force of gravity, is used to increase the sedimentation rate of solid sludge particles.
The most common centrifuge found in waste treatment dewatering applications is the continuous bowl centrifuge (Figure 37-14). The two principal elements of a continuous solid bowl centrifuge are the rotating bowl and the inner screw conveyor. The bowl acts as a settling vessel; the solids settle due to centrifugal force from its rotating motion. The screw conveyor picks up the solids and conveys them to the discharge port.
Often, operation of centrifugal dewatering equipment is a compromise between centrate quality, cake dryness, and sludge throughput. For example, an increase in solids throughput reduces clarification capacity, causing a decrease in solids capture. At the same time, the cake is drier due to the elimination of fine particles that become entrained in the centrate. The addition of polymers, with their ability to agglomerate fine particles, can result in increased production rates without a loss in centrate quality.
Polymers are usually fed inside the bowl because shear forces may destroy flocs if they are formed prior to entry. Also, large particles settle rapidly in the first stage of the bowl. Thus, economical solids recovery can be achieved through internal feeding of polymers after the large particles have settled.
Plate and Frame Press. A plate and frame filter press is a batch operation consisting of vertical plates held in a frame. A filter cloth is mounted on both sides of each plate. Sludge pumped into the unit is subjected to pressures of up to 25 psig as the plates are pressed together. As the sludge fills the chamber between individual plates, the filtrate flow ceases, and the dewatering cycle is completed. This cycle usually lasts from to 2 hr.
Because of the high pressures, blinding of the filter cloth by small sludge particles can occur. A filter precoat (e.g., diatomaceous earth) can be used to prevent filter blinding. Proper chemical conditioning of the sludge reduces or eliminates the need for precoat materials. At 5-10 psig, polymers can produce a rigid floc and eliminate fine particles. At greater pressures, the effectiveness of synthetic polymers is reduced; therefore, inorganic chemicals, such as ferric chloride and lime, are often used instead of polymers.
Sludge Drying Beds. Sludge drying beds consist of a layer of sand over a gravel bed. Underdrains spaced throughout the system collect the filtrate, which usually is returned to the wastewater plant.
Water is drained from the sludge cake by gravity through the sand and gravel bed. This process is complete within the first 2 days. All additional drying occurs by evaporation, which takes from 2 to 6 weeks. For this reason, climatic conditions, such as frequency and rate of precipitation, wind velocity, temperature, and relative humidity, play an important role in the operation of sludge drying beds. Often, these beds are enclosed to aid in dewatering. Chemical conditioning also reduces the time necessary to achieve the desired cake solids.
Sludge Disposal
Disposal of the sludge generated by wastewater treatment plants is dependent on government regulations (such as the Resource Conservation and Recovery Act), geographical location, and sludge characteristics, among other things. Final disposal methods include reclamation, incin-eration, land application, and landfill.
Reclamation. Because of costs associated with the disposal of wastewater sludge, each waste stream should be evaluated for its reclamation potential. Energy value, mineral content, raw material makeup, and by-product markets for each sludge should be evaluated. Examples include burning of digester gas to run compressors, recalcination of lime sludge to recover CaO, return of steel mill thickener sludge to the sinter plant, and marketing of by-product metallic salts for wastewater treatment use.
Incineration.Biological sludge can be disposed of by incineration; the carbon, nitrogen, and sulfur are removed as gaseous by-products, and the inorganic portion is removed as ash. Old landfill sites are filling up and new ones are becoming increasingly difficult to obtain. Therefore, waste reduction through incineration is becoming a favored disposal practice.
Several combustion methods are available, including hogged fuel boilers, wet air oxidation and kiln, multiple hearth furnace, and fluidized bed combustion processes.
Sludge incineration is a two-step process involving drying and combustion. Incineration of waste sludge usually requires auxiliary fuel to maintain temperature and evaporate the water contained in the sludge. It is critically important to maintain a low and relatively constant sludge moisture.
Land Application. Sludge produced from biological oxidation of industrial wastes can be used for land application as a fertilizer or soil conditioner. A detailed analysis of the sludge is important in order to evaluate toxic compound and heavy metal content, leachate quality, and nitrogen concentration.
Soil, geology, and climate characteristics are all important considerations in determining the suitability of land application, along with the type of crops to be grown on the sludge-amended soil. Sludge application rates vary according to all of these factors.
Landfill. Landfill is the most common method of industrial wastewater treatment plant sludge disposal.
Care must be taken to avoid pollution of groundwater. The movement and consequent recharge of groundwater is a slow process, so contamination that would be very small for a stream or river can result in irreversible long-term pollution of the groundwater. Many states require impermeable liners, defined as having a permeability of 10-7 cm/sec, in landfill disposal sites. This requirement limits liners to a few natural clays and commercial plastic liners. In addition to impermeable liners, leachate collection and treat-ment systems are typically required for new and remediated landfills.
Steps can be taken to reduce leachate and leachate contamination. Decreasing the moisture in the sludge removes water that would eventually be available as leachate. Proper consideration of the hydraulics of the landfill site can capture more rainfall as runoff and eliminate ponding and its contribution to leachate.
Many governmental regulations have been established in recent years for the protection of the environment. The Clean Water Act and the Resource Conservation and Recovery Act are among the most significant.
Clean Water Act
The Clean Water Act (CWA) of 1972 established regulations for wastewater discharge, provided funding for Publicly Owned Treatment Works (municipal waste treatment plants), and authorized the National Pollutant Discharge Elimination Systems (NPDES) to regulate and establish wastewater discharge permits for industrial and municipal plants.
Resource Conservation and Recovery Act (RCRA)
The Resource Conservation and Recovery Act (RCRA) of 1976 provided regulations for management of hazardous solid wastes, cleanup of hazardous waste sites, waste minimization, underground storage, and groundwater monitoring.
Learn more about Veolia's various wastewater treatment products and services.
Figure 37-1. Wastewater requires proper treatment before it is discharged from a plant.

Figure 37-2. Water consumption can be reduced through recycling and reuse of wastewater.

Figure 37-3. Circular clarifiers are used for mechanical removal of settleable solids from waste. (Reprinted with permission from Power.)
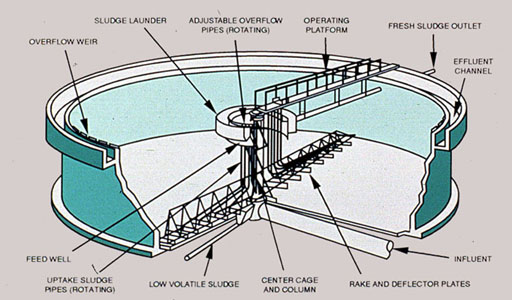
Figure 37-4. Proper pH adjustment is critical for optimum precipitation of metals.
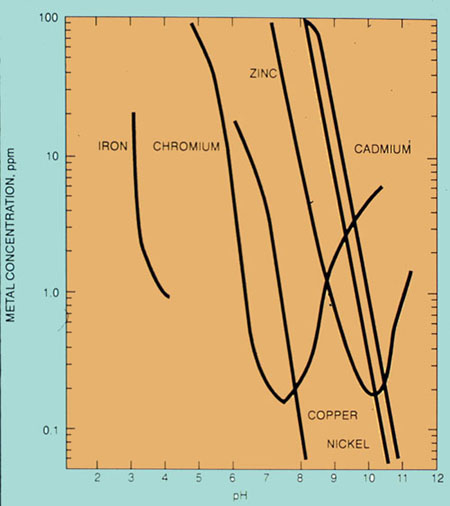
Figure 37-5. Biological oxidation converts soluble waste into clean water and an insoluble biomass.
Figure 37-6. Mechanical aeration provides oxygen for increased bacterial metabolism of dissolved organic pollutants in an activated sludge plant.

Figure 37-7. Activated sludge process returns active biomass to enhance waste removal.
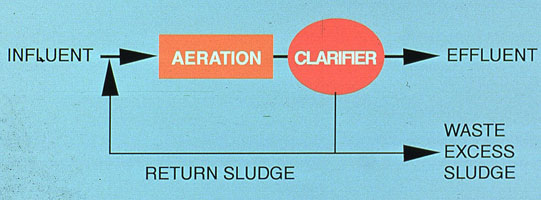
Figure 37-8. Model of bacterial population as a function of time and amount of food.
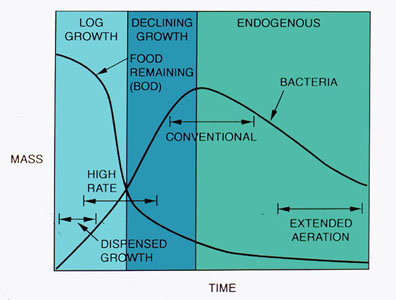
Figure 37-9. Odor control and solids reduction are accomplished in a digester.
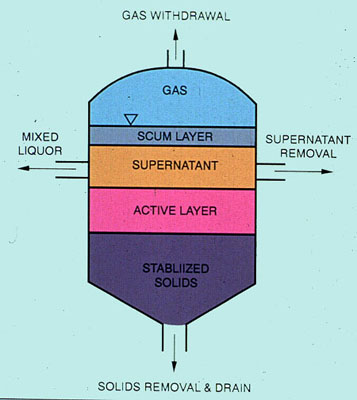
Figure 37-10. Solid waste volume is drastically reduced when water is removed.

Figure 37-11. Unconditioned sludge can be difficult to dewater.
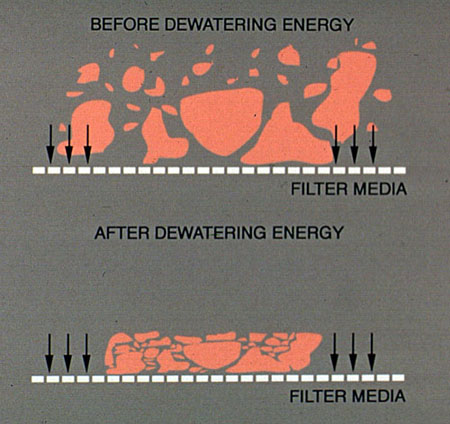
Figure 37-12. Gravity drainage is an important step in belt press dewatering.
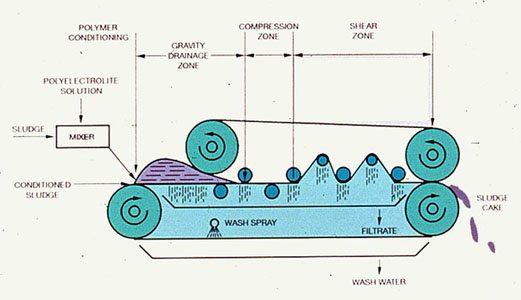
Figure 37-13. Screw presses are making inroads in the pulp and paper industry.
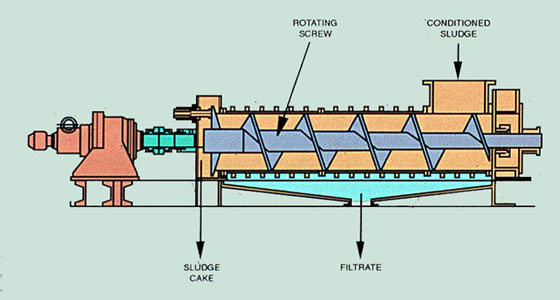
Figure 37-14. Proper adjustments for cake dryness and centrate quality are key to the efficient operation of a continuous bowl centrifuge.
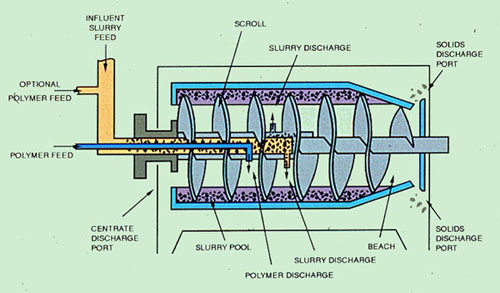
Table 37-1. Factors affecting biological oxidation.
| Food, BOD | To maintain control with efficient BOD removal, the proper amount of food must be supplied. |
| Dissolved oxygen | Insufficient oxygen levels inhibit BOD removal |
| pH/toxicants | With time, bacteria adapt to changes in conditions. Rapid changes in pH or type of waste organic inhibit the process. |
| Time | The degree of degradation varies with time. |
| Nutrients | Bacteria require trace amounts of ntirogen and phosphorus for cell maintenance. |
| Temperature | Low temperatures slow reaction rates; higher temperatures kill many strains of bacteria. |
Table 37-2 Typical removal efficiencies for oil refinery treatment processes.
|
|
Removal Efficiency, % |
||||||||
| Process | Process Influent | BOD | COD | TOC | SS | Oil | Phenol | Ammonia | Sulfide |
| API Separator | raw waste | 5-40 | 5-30 | NA | 10-50 | 60-99 | 0-50 | NA | NA |
| Primary clarifier | API effluent | 30-60 | 20-50 | NA | 50-80 | 60-95 | 0-50 | NA | NA |
| Dissolved air flotation | separator effluent | 20-70 | 10-60 | NA | 80-85 | 70-85 | 10-75 | NA | NA |
| Filter | API effluent | 40-70 | 20-55 | NA | 75-95 | 65-90 | 5-20 | NA | NA |
| Secondary oxidation pond | API effluent | 40-95 | 30-65 | 60 | 20-70 | 50-90 | 60-99 | 0-15 | 70-100 |
| Aerated lagoon | primary effluent | 75-95 | 60-85 | NA | 40-65 | 70-90 | 90-99 | 10-45 | 95-100 |
| Activated sludge | primary effluent | 80-99 | 50-95 | 40-90 | 60-85 | 80-99 | 95-99 | 33-99 | 97-100 |
| Trickling filter | API effluent | 60-85 | 30-70 | NA | 60-85 | 50-80 | 70-98 | 15-90 | 70-100 |
| Cooling tower | primary effluent | 50-95 | 40-90 | 10-70 | 50-85 | 60-75 | 75-99+ | 60-95 | NA |
| Activated carbon | primary effluent | 70-95 | 70-90 | 50-80 | 60-90 | 75-95 | 90-100 | 7-33 | NA |
| Tertiary filter granular media | secondary effluent | NA | NA | 50-65 | 75-95 | 65-95 | 5-20 | NA | NA |
| Activated carbon | secondary + filter effluent |
91-98 | 86-94 | 50-80 | 60-90 | 70-95 | 90-99 | 33-87 | NA |
Table 37-3 Activated sludge
| Aeration Retention Time, hr | MLSS, ppm | Aeration D.O., ppm | Sludge Recycle, % | BOD Loading, lb/Mft3 | F/M lb BOD/lb MLVSS | Sludge Production, lb/lb of BOD | BOD Removal % | |
| High rate | ½-3 | 300-1,000 | 0.5-2.0 | 5-15 | 2.5 | 1.5-5.0 | 0.65-0.85 | 75-85 |
| Conventional activated sludge | 6-8 (diffused) | 1,000-3,000 | 0.5-2.0 | 20-30 | 20-40 | 0.2-0.5 | 0.35-0.55 | 85-90 |
| 9-12 (mechanical) | 500-1,500 | 0.5-2.0 | 10-20 | 20-40 | 0.2-0.5 | 0.35-0.55 | 80-95 | |
| Extended aeration | 18-36 | 3,000-6,000 | 0.5-2.0 | 75-100 | 10-25 | 0.03-0.15 | 0.15-0.20 | 90-95 |
| Step aeration | 3-5 | 2,000-3,500 | 0.5-2.0 | 25-75 | 40-60 | 0.2-0.5 | 0.35-0.55 | 85-90 |
| Contact stabilization | 3-6 | 1,000-3,000 (aeration) | 0.5-2.0 | 25-100 | 60-75 | 0.2-0.6 | 0.35-0.55 | 85-90 |
| 4,000-10,000 (contact basin) | 0.5-2.0 | 25-100 | 60-75 | 0.2-0.6 | 0.35-0.55 | 85-90 | ||
| Pure Oxygen | 1-3 | 3,000-8,000 | 2-6 | 25-50 | 100-250 | 0.25-1.0 | 0.35-0.55 | 95-98 |
| Complete mix | 3-5 | 3,000-6,000 | 0.5-2.0 | 25-100 | 50-120 | 0.2-0.6 | 0.35-0.55 | 85-95 |
