- Oxidizing Antimicrobials in Industrial Cooling Systems
- Gaseous Chlorine Alternatives
- Bromine
- Halogen Donors
- Chlorine Dioxide
- Ozone
- Dechlorination
- Other Uses and Effects of Chlorine
- Feed Equipment
Chlorine is one of the most versatile chemicals used in water and wastewater treatment. This powerful oxidizing agent is used for:
- Disinfection
- control of microorganisms
- removal of ammonia
- control of taste and odor
- color reduction
- destruction of organic matter
- hydrogen sulfide oxidation
- iron and manganese oxidation
Although chlorine is beneficial for many uses, its use carries safety and environmental concerns.
Physical Properties and Aqueous Reactions
Chlorine in its gaseous state was discovered by Karl W. Scheele in 1774 and identified as an element by Humphrey Davy in 1810. Chlorine gas is greenish-yellow, and its density is about 2 times that of air. When condensed, it becomes a clear, amber liquid with a density about 1 times greater than water. One volume of liquid chlorine yields approximately 500 volumes of chlorine gas, which is neither explosive nor flammable. Like oxygen, chlorine gas can support the combustion of some substances. Chlorine reacts with organic materials to form oxidized or chlorinated derivatives. Some of these reactions, such as those with hydrocarbons, alcohols, and ethers, can be explosive. The formation of other chlorinated organics, specifically trihalomethanes (THM), poses an environmental threat to public drinking water supplies.
Chlorine gas is also a toxic respiratory irritant. Airborne concentrations greater than 3-5 ppm by volume are detectable by smell, and exposure to 4 ppm for more than 1 hr can have serious respiratory effects. Because chlorine gas is denser than air, it stays close to the ground when released. The contents of a 1-ton cylinder of chlorine can cause coughing and respiratory discomfort in an area of 3 square miles. The same amount concentrated over an area of 1/10 square mile can be fatal after only a few breaths.
Chlorine is generated commercially by the electrolysis of a brine solution, typically sodium chloride, in any of three types of cells: diaphragm, mercury, or membrane. The majority of chlorine produced in the United States is manufactured by the electrolysis of sodium chloride to form chlorine gas and sodium hydroxide in diaphragm cells. The mercury cell process produces a more concentrated caustic solution (50%) than the diaphragm cell. Chlorine gas can also be generated by the salt process (which employs the reaction between sodium chloride and nitric acid), by the hydrochloric acid oxidation process, and by the electrolysis of hydrochloric acid solutions. The gas is shipped under pressure in 150-lb cylinders, 1-ton cylinders, tank trucks, tank cars, and barges.
The four basic categories of chlorine treatment are defined not only by their function but also by their position in a water treatment sequence:
- prechlorination
- rechlorination
- post-chlorination
- dechlorination
In chemically pure water, molecular chlorine reacts with water and rapidly hydrolyzes to hypochlorous acid (HOCl) and hydrochloric acid (HCl):
|
Cl2
|
+
|
H2O
|
®
|
HOCl
|
+
|
HCl
|
|
chlorine
|
|
water
|
|
hypochlorous
acid |
|
hydrochloric
acid |
Both of the acids formed by hydrolysis react with alkalinity to reduce buffering capacity of water and lower pH. Every pound of chlorine gas added to water removes about 1.4 lb of alkalinity. In cooling water systems, this alkalinity reduction can have a major impact on corrosion rates.
At pH levels above 4.0 and in dilute solutions, the hydrolysis reaction is completed within a fraction of a second. For all practical purposes the reaction is irreversible. Hypochlorous acid is a weak acid and dissociates to form a hydrogen ion and a hypochlorite ion.
| HOCl | « » | H+ | + | OCl |
| hypochlorous acid |
hydrogen ion |
hypochlorite ion |
The concentration or distribution of each species at equilibrium depends on pH and temperature. Between pH 6.5 and 8.5, the dissociation reaction is incomplete, and both hypochlorous acid and hypochlorite ions are present. The equilibrium ratio at any given pH remains constant even if the hypochlorous acid concentration is decreasing. At constant pH and increasing temperature, chemical equilibrium favors the OCl - ion over HOCl.
The primary oxidizing agents in water are hypochlorous acid and the hypochlorite ion, although hypochlorite has a lower oxidizing potential. Oxidizing potential is a measure of the tendency of chlorine to react with other materials. The speed at which these reactions occur is determined by pH, temperature, and oxidation/reduction potential. As pH increases, the chemical reactivity of chlorine decreases; as temperature increases, reactions proceed more rapidly. The oxidation reactions of chlorine with such inorganic reducing agents as sulfides, sulfites, and nitrites are generally very rapid. Some dissolved organic materials also react rapidly with chlorine, but the completion of many organic-chlorine reactions can take hours.
Chlorine Demand. Chlorine Demand. Chlorine demand is defined as the difference between the amount of chlorine added to a water system and the amount of free available chlorine or combined available chlorine remaining at the end of a specified time period. The demand is the amount of chlorine consumed by oxidation or substitution reactions with inorganic and organic materials, such as H2S, Fe2+, Mn2+, NH3, phenols, amino acids, proteins, and carbohydrates. Free available residual chlorine is the amount of chlorine which exists in the treated water system as hypochlorous acid and hypochlorite ions after the chlorine demand has been satisfied. Free residual chlorination is the application of chlorine to water to produce a free available chlorine residual.
Combined Available Residual Chlorination. Combined Available Residual Chlorination. Combined available residual chlorine is the chlorine residual which exists in water in combination with ammonia or organic nitrogen compounds. Combined residual chlorination is the application of chlorine to water to react with ammonia (natural or added) or other nitrogen compounds to produce a combined available chlorine residual. Total available chlorine is the total of free available chlorine, combined available chlorine, and other chlorinated compounds.
Available Chlorine. Available Chlorine. "Available chlorine" is an expression of the equivalent weights of oxidizing agents, with chlorine gas as a basis, similar to the expression of alkalinity in terms of calcium carbonate equivalents. The term originated from the need to compare other chlorine-containing compounds to gaseous chlorine. Available chlorine is based on the half-cell reaction in which chlorine gas is reduced to chloride ions with the consumption of two electrons. In this reaction, the equivalent weight of chlorine is the molecular weight of chlorine, 71 g/mole, divided by 2, or 35.5 g/mole.
|
2e -
|
+
|
Cl2
|
®
|
2Cl -
|
|
|
|
chlorine
|
|
chloride ion
|
The available chlorine of other chlorine-containing compounds is calculated from similar half-cell reactions, the formula weight of the compound, and the equivalent weight of chlorine.
Even though chlorine gas only dissociates into 50% HOCl or OCl -, it is considered 100% available chlorine. Because of this definition, it is possible for a compound to have more than 100% available chlorine. The active weight percent chlorine multiplied by 2 indicates available chlorine. Table 27-1 lists actual weight percent and available chlorine percent for several common compounds.
Available chlorine, like oxidation potential, is not a reliable indicator of the occurrence or extent of an oxidation reaction. It is an even poorer indicator of the antimicrobial efficacy of an oxidizing compound. For example, the antimicrobial efficacy of hypochlorous acid (HOCl) is much greater than any of the chloramines even though the chloramines have a higher available chlorine.
Chloramine Formation. Chloramine Formation. One of the most important reactions in water conditioning is the reaction between dissolved chlorine in the form of hypochlorous acid and ammonia (NH3) to form inorganic chloramines. The inorganic chloramines consist of three species: monochloramine (NH2Cl), dichloramine (NHCl2), and trichloramine, or nitrogen trichloride (NCl3). The principal reactions of chloramine formation are:
| NHCl2 | + | HOCl | ® | NCl3 | + | H2O |
| dichloramine | hypochlorous acid |
trichloramine | water |
| NH3 (aq) | + | HOCl | ® | NH2Cl | + | H2O |
| ammonia | hypochlorous acid |
monochloramine | water |
| NH2Cl | + | HOCl | ® | NHCl2 | + | H2O |
| monochloramine | hypochlorous acid |
dichloramine | water |
The relative amounts of chloramines formed are a function of the amount of chlorine fed, the chlorine/ammonia ratio, temperature, and pH. In general, monochloramine is formed above pH 7 and predominates at pH 8.3. Dichloramine predominates at pH 4.5. Between these pH values, mixtures of the two chloramines exist. Below pH 4.5, nitrogen trichloride is the predominant reaction product.
The oxidizing potential of monochloramines is substantially lower than that of chloride, and monochloramines are slower to react with organic matter. These properties reduce the amount of trihalomethanes (THM) formed. The formation of THM is considered more detrimental in potable water than the reduction of the antimicrobial capabilities of free chlorine. Therefore, ammonia is often injected into the chlorine feed stream to form chloramines before the chlorine is fed into the potable water stream.
Combined chlorine residuals are ordinarily more chemically stable (less reactive with chlorine demand) than free chlorine residuals. This property helps maintain stable residuals in outlying pressurized water distribution systems. However, the lower antimicrobial effectiveness of chloramines compared to free chlorine requires higher combined residuals and/or longer contact times, which are often available in distribution systems.
Breakpoint Chlorination. Breakpoint chlorination is the application of sufficient chlorine to maintain a free available chlorine residual. The principal purpose of breakpoint chlorination is to ensure effective disinfection by satisfying the chlorine demand of the water. In wastewater treatment, breakpoint chlorination is a means of eliminating ammonia, which is converted to an oxidized volatile form.
The addition of chlorine to a water that contains ammonia or nitrogen-containing organic matter produces an increased combined chlorine residual. Mono- and dichloramines are formed between points A and B on this curve. After the maximum combined residual is reached (point B), further chlorine doses decrease the residual. Chloramine oxidation to dichloramine, occurring between points B and C, results in a decline in the combined available residuals initially formed. Point C represents the breakpoint: the point at which chlorine demand has been satisfied and additional chlorine appears as free residuals. Between points C and D, free available residual chlorine increases in direct proportion to the amount of chlorine applied.
Factors that affect breakpoint chlorination are initial ammonia nitrogen concentration, pH, temperature, and demand exerted by other inorganic and organic species. The weight ratio of chlorine applied to initial ammonia nitrogen must be 8:1 or greater for the breakpoint to be reached. If the weight ratio is less than 8:1, there is insufficient chlorine present to oxidize the chlorinated nitrogen compounds initially formed. When instantaneous chlorine residuals are required, the chlorine needed to provide free available chlorine residuals may be 20 or more times the quantity of ammonia present. Reaction rates are fastest at pH 7-8 and high temperatures.
On a typical breakpoint curve the initial chlorine dosage produces no residual because of an immediate chlorine demand caused by fast-reacting ions. As more chlorine is applied, chloramines develop. These chloramines are shown in the total chlorine residual. At higher chlorine dosages, the slope to breakpoint begins. After the breakpoint, free chlorine residuals develop.
Free chlorine residuals usually destroy tastes and odors, control exposed bacteria, and oxidize organic matter. Breakpoint chlorination can also control slime and algae growths, aid coagulation, oxidize iron and manganese, remove ammonia, and generally improve water quality in the treatment cycle or distribution system.
OXIDIZING ANTIMICROBIALS IN INDUSTRIAL COOLING SYSTEMS
The oxidizing antimicrobials commonly used in industrial cooling systems are the halogens, chlorine and bromine, in liquid and gaseous form; organic halogen donors; chlorine dioxide; and, to a limited extent, ozone.
Oxidizing antimicrobials oxidize or accept electrons from other chemical compounds. Their mode of antimicrobial activity can be direct chemical degradation of cellular material or deactivation of critical enzyme systems within the bacterial cell. An important aspect of antimicrobial efficiency is the ability of the oxidizing agent to penetrate the cell wall and disrupt metabolic pathways. For this reason, oxidation potential alone does not always correlate directly with antimicrobial efficiency.
The relative microbiological control ability of typical halogens is as follows:
| HOCl | 3 | HOBr | 3 | NHxBry | >> |
| hypochlorous acid | hypobromous acid | bromamine |
| OCl - | > | OBr - | >>> | NHxCly | |
| hypochlorite ion | hypobromite ion | chloramine |
Cooling water pH affects oxidizing antimicrobial efficacy. pH determines the relative proportions of hypochlorous acid and hypochlorite ion or, in systems treated with bromine donors, hypobromous acid and hypobromite ion. The acid forms of the halogens are usually more effective antimicrobials than the dissociated forms. Under some conditions, hypochlorous acid is 80 times more effective in controlling bacteria than the hypochlorite ion. Hypochlorous acid predominates below a pH of 7.6. Hypobromous acid predominates below pH 8.7, making bromine donors more effective than chlorine donors in alkaline cooling waters, especially where contact time is limited.
Antimicrobial efficacy is also affected by demand in the cooling water system, specifically demand exerted by ammonia. Chlorine reacts with ammonia to form chloramines, which are not as efficacious as hypochlorous acid or the hypochlorite ion in microbiological control. Bromine reacts with ammonia to form bromamines. Unlike chloramines, bromamines are unstable and reform hypobromous acid.
Most microbes in cooling systems can be controlled by chlorine or bromine treatment if exposed to a sufficient residual for a long enough time. A free chlorine residual of 0.1-0.5 ppm is adequate to control bulk water organisms if the residual can be maintained for a sufficient period of time.
Continuous chlorination of a cooling water system often seems most prudent for microbial slime control. However, it is economically difficult to maintain a continuous free residual in some systems, especially those with process leaks. In some high-demand systems it is often impossible to achieve a free residual, and a combined residual must be accepted. In addition, high chlorine feed rates, with or without high residuals, can increase system metal corrosion and tower wood decay. Supplementing with nonoxidizing antimicrobials is preferable to high chlorination rates.
In once-through systems, free residuals from 0.3 to 0.8 ppm are normally maintained for -2 hr per treatment period. The rate of recontamination determines the frequency of treatment required.
Open recirculating systems can be treated with a continuous or intermittent halogenation program. Continuous feed is the most effective and is generally affordable where chlorine gas or hypochlorite is being used and system demand is low. Free residuals of 0.1-0.5 ppm are manually maintained. Care should be taken not to feed excessive amounts of halogen that will adversely affect corrosion rates. Chlorine feed rates should not exceed 4 ppm based on recirculation rate. The use of halogen donors may be restricted to intermittent feed due to economics, although continuous feed in low-demand systems is effective. Intermittent feed requires maintaining the same free residual as in the continuous program but for only the last to 1 hr of the chlorine application. Up to 3 hr of chlorine addition may be required in order to achieve the free residual, depending on system demand, system cleanliness, and the frequency of chlorination.
Community right-to-know laws, Superfund Reauthorization, SARA Title III laws, and the release of a deadly gas in Bhopal, India, have raised serious concerns regarding the safety of gaseous chlorine. Other sources of halogens and oxidizing agents for microbiological control include:
- hypochlorites (sodium hypochlorite, sodium hypochlorite with sodium bromide, and calcium hypochlorite)
- chlorinated or brominated donor molecules, such as isocyanurates, trichloro-s-triazinetriones, and hydantoins
- chlorine dioxide
- ozone
Hypochlorites
Sodium hypochlorite and calcium hypochlorite are chlorine derivatives formed by the reaction of chlorine with hydroxides. The application of hypochlorite to water systems produces the hypochlorite ion and hypochlorous acid, just as the application of chlorine gas does.
| NaOCl | ® | OCl - | + | Na+ |
| sodium hypochlorite | hypochlorite ion | sodium ion |
| OCl - | + | Na+ | + | H2O | « | HOCl | + | NaOH |
| hypochlorite ion | sodium ion | water | hypochlorous acid | sodium hydroxide |
| Ca(OCl)2 | ® | 2OCl | + | Ca2+ |
| calcium hypochlorite | hypochlorite ion | calcium ion |
|
2OCl -
|
+
|
Ca2+
|
+
|
2H2O
|
« »
|
2HOCl
|
+
|
Ca(OH)2
|
|
hypochlorite ion
|
|
calcium ion
|
|
water
|
|
hypochlorous acid
|
|
calcium hydroxide
|
The difference between the hydrolysis reaction of chlorine gas and hypochlorites is the reaction by-products. The reaction of chlorine gas and water increases the H+ ion concentration and decreases pH by the formation of hydrochloric acid. The reaction of hypochlorites and water forms both hypochlorous acid and sodium hydroxide or calcium hydroxide. This causes little change in pH. Solutions of sodium hypochlorite contain minor amounts of excess caustic as a stabilizer, which increase alkalinity and raise pH at the point of injection. This can cause hardness scale to form. Addition of a dispersant (organic phosphate/polymer) to the water system is usually sufficient to control this scaling potential.
Alkalinity and pH are significantly changed when sodium or calcium hypochlorite replaces gaseous chlorine. Gaseous chlorine reduces alkalinity by 1.4 ppm per ppm of chlorine fed; hypochlorite does not reduce alkalinity. The higher alkalinity of waters treated with hypochlorite reduces the corrosion potential but can increase the deposition potential.
Sodium Hypochlorite. Sodium Hypochlorite. Sodium hypochlorite, also referred to as liquid bleach, is the most widely used of all the chlorinated bleaches. It is available in several solution concentrations, ranging from the familiar commercial variety at a concentration of about 5.3 weight percent NaOCl to industrial strengths at concentrations of 10-12%. The strength of a bleach solution is commonly expressed in terms of its "trade percent" or "percent by volume," not its weight percent: 15 trade percent hypochlorite is only 12.4 weight percent hypochlorite. Approximately 1 gal of industrial strength sodium hypochlorite is required to replace 1 lb of gaseous chlorine.
The stability of hypochlorite solutions is adversely affected by heat, light, pH, and metal contamination. The rate of decomposition of 10% and 15% solutions nearly doubles with every 10°F rise in the storage temperature. Sunlight reduces the half-life of a 10%-15% hypochlorite solution by a factor of 3 to 5. If the pH of a stored solution drops below 11, decomposition is more rapid. As little as 0.5 ppm of iron causes rapid deterioration of 10-15% solutions. The addition of concentrated ferric chloride to a tank of sodium hypochlorite causes the rapid generation of chlorine gas.
Normal industrial grades of sodium hypochlorite may be fed neat or diluted with low-hardness water. The use of high-hardness waters for dilution can cause precipitation of calcium salts due to the high pH of the hypochlorite solution.
"High Test" Calcium Hypochlorite (HTH). "High Test" Calcium Hypochlorite (HTH). The most common form of dry hypochlorite in the United States is high test calcium hypochlorite (HTH). It contains 70% available chlorine, 4-6% lime, and some calcium carbonate. Precipitates form when HTH is dissolved in hard water. For feeding calcium hypochlorite as a liquid, solutions should be prepared with soft water at 1-2% chlorine concentration. Care should be exercised in storing granular calcium hypochlorite. It should not be stored where it may be subjected to heat or contacted by easily oxidized organic material. Calcium hypochlorite decomposes exothermally, releasing oxygen and chlorine monoxide. Decomposition occurs if HTH is contaminated with water or moisture from the atmosphere. Calcium hypochlorite loses 3-5% of its chlorine content per year in normal storage.
All hypochlorites are somewhat harmful to skin and must be handled carefully. Corrosion-resistant materials should be used for storing and dispensing.
Bromine has been used for water treatment since the 1930's. Most bromine production in the United States occurs in the Great Lakes region and Arkansas. Bromine is generated commercially through the reaction of a bromine brine solution with gaseous chlorine, followed by stripping and concentration of the bromine liquid. Bromine is a fuming, dark red liquid at room temperature.
Bromine dissociates in water in the same manner as chlorine, by forming hypobromous acid and the hypobromite ion. Hypobromous acid is a weak acid that partially dissociates to form a hydrogen ion and a hypobromite ion. The concentration or distribution of each species at equilibrium depends on pH and temperature. Between pH 6.5 and 9, the dissociation reaction is incomplete, and both hypobromous acid and the hypobromite ion are present. The equilibrium ratio at any given pH remains constant. At a pH above 7.5, the amount of hypobromous acid is greater than the amount of hypochlorous acid for equivalent feed rates. The higher percentage of hypobromous acid is beneficial in alkaline waters and in ammonia-containing waters.
Methods of generating hypobromous acid include:
using two liquids (or one liquid and chlorine gas)
| NaBr | + | HOCl | ® | HOBr | + | NaCl |
| sodium bromide | hypochlorous acid | hypobromous acid | salt |
using a compressed gas
| BrCl | + | H2O | ® | HOBr | + | HCl |
| bromine chloride | water | hypobromous acid | hydrochloric acid |
using a solid
| C5H6BrClN2O2 | + | 2H2O | ® | HOCl | + | HOBr | + | C5H8N2O2 |
| bromochloro- dimethylhydantoin (BCDMH) |
water | hypochlorous acid | hypobromous acid | dimethyl- hydantoin |
Regardless of the method used to generate hypobromous acid, the goal is to take advantage of its antimicrobial activity. The liquid and solid methods do not require the storage of compressed gases-the major reason for gaseous chlorine replacement.
Bromine reacts with ammonia compounds to form bromamines, which are much more effective antimicrobials than chloramines. At pH 8.0, the hypobromous acid to bromamine ratio is 8:1 in ammonia-containing waters. Because monobromamine is unstable and because tribromamine is not formed, there is little need to proceed to breakpoint bromination.
The shorter life expectancy of bromine compounds (due to lower bond strength) lowers oxidizer residuals in plant discharges and reduces the need to dechlorinate before discharge.
Halogen donors are chemicals that release active chlorine or bromine when dissolved in water. After release, the halogen reaction is similar to that of chlorine or bromine from other sources. Solid halogen donors commonly used in cooling water systems include the following:
- 1-bromo-3-chloro-5,5-dimethylhydantoin
- 1,3-dichloro-5,5-dimethylhydantoin
- sodium dichloroisocyanurate
These donor chemicals do not release the active halogen all at once, but make it slowly available; therefore, they may be considered "controlled release" oxidizing agents. Their modes of action are considered to be similar to chlorine or bromine, but they can penetrate cell membranes and carry out their oxidative reactions from within the cell. These donors are being widely used because of the simplicity, low capital cost, and low installation cost of the feed systems. In addition, because they are solids, they eliminate the handling hazards associated with gases (escapement) and liquids (spills). Evaluated on a total cost basis, halogen donors often prove to be an economical choice despite their relatively high material costs.
Chlorine dioxide, ClO2, is another chlorine derivative. This unstable, potentially explosive gas must be generated at the point of application. The most common method of generating ClO2 is through the reaction of chlorine gas with a solution of sodium chlorite.
| 2NaClO2 | + | Cl2 | ® | 2ClO2 | + | 2NaCl |
| sodium chlorite | chlorine dioxide | chlorine | sodium chloride |
Theoretically, 1 lb of chlorine gas is required for each 2.6 lb of sodium chlorite. However, an excess of chlorine is often used to lower the pH to the required minimum of 3.5 and to drive the reaction to completion. Sodium hypochlorite can be used in place of the gaseous chlorine to generate chlorine dioxide. This process requires the addition of sulfuric or hydrochloric acid for pH control.
Other methods used for chlorine dioxide generation include:
| 5NaClO2 | + | 5HCl | ® | 4ClO2 | + | 5NaCl | + | HCl | + | 2H2O |
| sodium chlorite | hydrochloric acid | chlorine dioxide | sodium chloride | hydrochloric acid | water |
| 10NaClO2 | + | 5H2SO4 | ® | 8ClO2 | + | 5Na2SO4 | + | 2HCl | + | 4H2O |
| sodium chlorite | sulfuric acid | chlorine dioxide | sodium sulfate | hydrochloric acid | water |
|
2NaClO2
|
+
|
HCl
|
+
|
NaOCl
|
®
|
2ClO2
|
+
|
2NaCl
|
+
|
NaOH
|
|
sodium chlorite
|
|
hydrochloric acid
|
|
sodium hypochlorite
|
|
chlorine dioxide
|
|
sodium chloride
|
|
sodium hydroxide
|
Rather than hydrolyzing in water as chlorine does, chlorine dioxide forms a true solution in water under typical cooling tower conditions. For this reason, chlorine dioxide is volatile (700 times more volatile than HOCl) and may be easily lost from treated water systems, especially over cooling towers.
Chlorine dioxide is a powerful oxidant. It reacts rapidly with oxidizable materials but, unlike chlorine, does not readily combine with ammonia. Chlorine dioxide does not form trihalomethanes (THM) but can significantly lower THM precursors. In sufficient quantity, chlorine dioxide destroys phenols without creating the taste problems of chlorinated phenols. It is a good antimicrobial and antispore. Unlike chlorine, the antimicrobial efficiency of chlorine dioxide is relatively unaffected by changes in pH in the range of 6-9. Chlorine dioxide is also used for the oxidation of sulfides, iron, and manganese.
Complex organic molecules and ammonia are traditional chlorine-demand materials that do not react with chlorine dioxide. Because chlorine dioxide reacts differently from chlorine, a chlorine dioxide demand test must be conducted to determine chlorine dioxide feed rates. A residual must be maintained after the chlorine dioxide demand has been met, to ensure effective control of microbiological growth. The chemical behavior and oxidation characteristics of aqueous chlorine dioxide are not well understood because of the difficulty in differentiating aqueous chlorine-containing species.
Chlorine dioxide is applied to some public water supplies to control taste and odor, and as a disinfectant. It is used in some industrial treatment processes as an antimicrobial. Chlorine dioxide consumed in water treatment reactions reverts to chlorite ions (ClO2-), chlorate ions (ClO3- ), and chloride ions (Cl -). There are some concerns about the long-term health effects of the chlorite ion in potable water supplies.
As a gas, chlorine dioxide is more irritating and toxic than chlorine. Chlorine dioxide in air is detectable by odor at 14-17 ppm, irritating at 45 ppm, fatal in 44 min at 150 ppm, and rapidly fatal at 350 ppm. Concentrations greater than 14% in air can sustain a decomposition wave set off by an electric spark. The most common precursor for on-site generation of chlorine dioxide is also a hazardous material: liquid sodium chlorite. If allowed to dry, this powerful oxidizing agent forms a powdered residue that can ignite or explode if contacted by oxidizable materials. The hazardous nature of chlorine dioxide vapor and its precursor, and the volatility of aqueous solutions of chlorine dioxide, require caution in the design and operation of solution and feeding equipment.
Ozone is an allotropic form of oxygen, O3. Because it is an unstable gas, it must be generated at the point of use. Ozone is a very effective, clean oxidizing agent possessing powerful antibacterial and antiviral properties.
Because ozone is a strong oxidizing agent, it is a potential safety hazard. It has been reported that concentrations of 50 ppm of ozone in the air can cause oxidization of the lining of the lungs and accumulation of fluid, resulting in death by pulmonary edema. OSHA and NIOSH consider 10 ppm immediately dangerous to life or health, and the OSHA exposure limit is a time-weighted average of 0.1 ppm. In concentrations as low as 0.02 ppm, strong ozone odors are detectable. Improper operation of ozone-generating equipment can produce 20% ozone, an explosive concentration. Ozone-generating equipment must have a destruct mechanism to prevent the release of ozone to the atmosphere where it can cause the formation of peroxyacetyl nitrate (PAN), a known air pollutant.
Ozone's short half-life may allow treated water to be discharged without harm to the environment. However, the shorter half-life reduces contact in a treated water system, so the far reaches of a wa-ter system may not receive adequate treatment.
Ozone is generated by dry air or oxygen being passed between two high-voltage electrodes. Ozone can also be generated photochemically by ultraviolet light. Ozone must be delivered to a water system by injection through a contactor. The delivery rate is dependent on the mass transfer rate of this contactor or sparger. Proper maintenance of the generator and contactor is critical.
High capital costs limit the use of ozone for microbiological growth control, particularly in systems with varying demand.
Dechlorination is often required prior to discharge from the plant. Also, high chlorine residuals are detrimental to industrial systems, such as ion exchange resins, and some of the membranes used in electrodialysis and reverse osmosis units. Chlorine may also contribute to effluent toxicity; therefore, its concentration in certain aqueous discharges is limited.
Sometimes, dechlorination is required for public and industrial water supplies. Reducing or removing the characteristic "chlorine" taste from potable water is often desirable. Dechlorination is commonly practiced in the food and beverage processing industries. Direct contact of water containing residual chlorine with food and beverage products is avoided, because undesirable tastes can result.
Excess free residual chlorine can be lowered to an acceptable level by chemical reducing agents, carbon adsorption, or aeration.
Chemical reducing agents, such as sulfur dioxide, sodium sulfite, and ammonium bisulfite, dechlorinate water but can also promote the growth of bacteria that metabolize sulfur. Sometimes, sodium thiosulfate is used to dechlorinate water samples prior to bacteriological analysis. Common dechlorination reactions are:
|
SO2 |
+ |
Cl2 |
+ |
2H2O |
« » |
H2SO4 |
+ |
2HCl |
|
sulfur dioxide |
chlorine |
water |
sulfuric acid |
hydrochloric acid |
| NaHSO3 | + | Cl2 | + | H2O | « » | NaHSO4 | + | 2HCl |
| sodium bisulfite | chlorine | water | sodium bisulfate | hydrochloric acid |
|
NH4HSO3 |
+ |
Cl2 |
+ |
H2O |
« » |
NH4HSO4 |
+ |
2HCl |
|
ammonium bisulfite |
chlorine |
water |
ammonium bisulfate |
hydrochloric acid |
Granular activated carbon (GAC) removes free chlorine by adsorption. Free chlorine in the form of HOCl reacts with activated carbon to form an oxide on the carbon surface. Chloramines and chlorinated organics are adsorbed more slowly than free chlorine.
Aeration is the least effective means of dechlorination, with effectiveness decreasing with increasing pH. The hypochlorite ion, which predominates at pH 8.3 and higher, is less volatile than hypochlorous acid.
Ultraviolet radiation dechlorinates water stored in open reservoirs for prolonged periods.
OTHER USES AND EFFECTS OF CHLORINE
In addition to serving as antimicrobials, chlorine and chlorine compounds are used to reduce objectionable tastes and odors in drinking water; improve influent clarification processes; oxidize iron, manganese, and hydrogen sulfide to facilitate their removal; reduce sludge bulking in wastewater treatment plants; and treat wastewater plant effluents.
Chlorine, along with a coagulant, is often applied to raw water in influent clarification processes. This prechlorination improves coagulation because of the effect of chlorine on the organic material in the water. It is also used to reduce taste, odor, color, and microbiological populations, and it oxidizes iron and manganese to facilitate removal by settling and filtration. One part per million of chlorine oxidizes 1.6 ppm of ferrous ion or 0.77 ppm of manganous ion. The addition of 8.87 ppm of chlorine per ppm of sulfide oxidizes sulfides to sulfates, depending on pH and temperature.
Chlorine is a successful activating agent for sodium silicate in the preparation of the coagulant aid, activated silica. The advantage of this process is that the chlorine used for activation is available for other purposes.
Low-level, intermittent chlorination of return activated sludge has been used to control severe sludge bulking problems in wastewater treatment plants.
Chlorine, injected into sewage and industrial wastes before they are discharged, destroys bacteria and such chemicals as sulfides, sulfites, and ferrous iron. These chemicals react with and consume dissolved oxygen in the receiving body of water.
Chlorination equipment is commercially available for liquefied chlorine gas and solutions of sodium hypochlorite. Calcium hypochlorite is a solid and is usually added by shot feeding. The more recent solid halogen donors, such as 1-bromo-3-chloro-5,5-dimethylhydantoin, are fed with bypass dissolving feeders.
Liquefied chlorine gas is the least expensive form of chlorine and has generally been the antimicrobial of choice in the past. Because of the hazards of chlorine leakage, feed equipment is designed to maintain the chlorine gas below atmospheric pressure by operating under a vacuum.This causes any leaks to be directed into the feeding system rather than into the surrounding atmosphere. Maximum solubility is about 5000 ppm at the vacuum levels currently produced by chlorine injection equipment. Chlorinator manufacturers design equipment to limit the amount of chlorine in the feed system to 3500 ppm to prevent the release of chlorine gas at the point of application. Direct injection of chlorine without the use of an appropriate eductor can be disastrous.
Sodium hypochlorite feeders include metering pumps, flow-controlled rotometers, and computerized feed systems, such as the Betz PaceSetter (see Chapter 35). The hypochlorite storage system should be protected from direct sunlight and heat to prevent degradation. Selection of appropriate storage metals is also important to prevent degradation.
Solid halogen donors, such as hydantoins, triazinetriones, and isocyanurates, are available in tablet form and, sometimes, in granular form. The solids are typically dissolved in a bypass feederto regulate the dissolution rate, and the concentrated feeder effluent is applied at the appropriate point. The chemicals provided by these products are hypochlorous acid, hypobromous acid, or a combination of the two.
Figure 27-1. Dissociation of hypobromous and hypochlorous acid with changing pH.
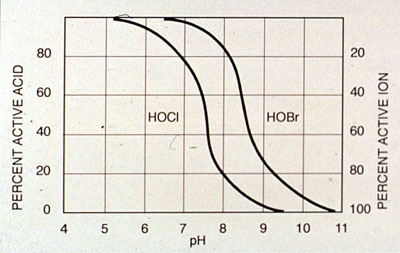
Figure 27-2. Theoretical breakpoint chlorination curve.
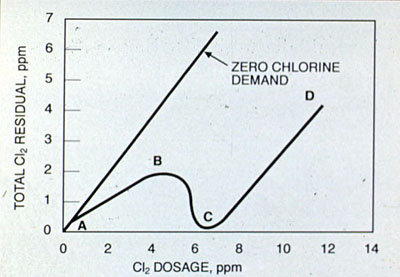
Figure 27-3. Typical breakpoint chlorination curve.
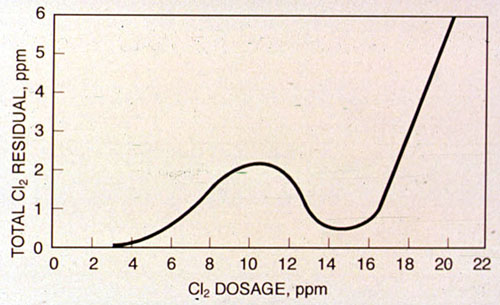
Figure 27-4. Flow Diagram for gas chlorination. (Courtesy of Capital Controls Company).
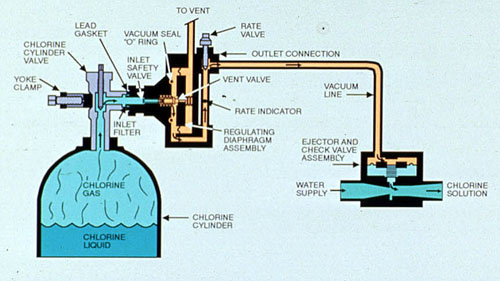
Figure 27-5. Solid Halogen Feeder.

