- Refrigeration Cycle
- Open Recirculating Cooling Water Problems
- Air Washers
- Closed Water Systems
- Control of Water Balances
- General Considerations
For more than a century, industrial air conditioning has been used for drying, humidity control, and dust and smoke abatement. Its most familiar function is to provide a comfortable working environment, to increase the comfort and productivity of personnel in offices, commercial buildings, and industrial plants.
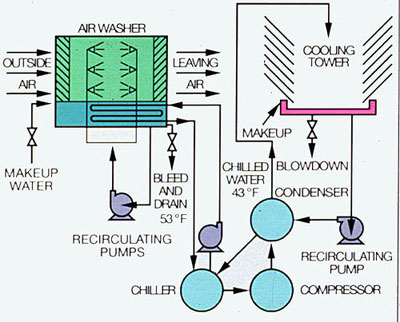
Air conditioning is the process of treating and distributing air to control temperature, humidity, and air quality in selected areas. For temperature and humidity control, air is moved over chilled or heated coils and/or a spray of water at a controlled temperature. Direct water sprays also remove dust and odors. Other air cleaning systems may include mechanical separation, adhesion, screening, filtration, or static attraction, depending on the type of air contaminants encountered and the required air quality(Figure 34-1).
Refrigeration is the process of lowering the temperature of a substance below that of its surroundings and includes production of chilled water for air conditioning or process applications. Chilled water for use in processes such as injection molding may be in the same temperature range as chilled water used for air conditioning. Refrigeration systems are also used to provide chilled antifreeze solutions (brines) at temperatures below the freezing point of water. Brines are used in icemaking and cold storage, in addition to a variety of chemical process applications.
Chilled water may be used in air washers, either in closed coils or as spray water. Chilled water may also be used for closed systems and for individual spray water systems.
Many methods are used to produce and distribute chilled air. In central air conditioning systems, air is passed over coils chilled by water, by brine, or by direct expansion of a volatile refrigerant. The chilled air is then distributed through ductwork.
The water systems associated with air conditioning can be classified into three general categories: open recirculating cooling, air washers, and closed or open chilled water systems. In water treatment applications, open recirculating cooling systems are similar to open chilled water systems.
The basic mechanical components of an air conditioning system are the air and water distribution systems, a refrigeration machine, and a heat rejection system. Refrigeration for air conditioning is usually provided by either absorption or compression cycles.
Absorption refrigeration uses low-pressure steam or high-temperature hot water as the energy source, water as the refrigerant, and lithium bromide or lithium chloride as the absorbent.
Compression refrigeration systems generally utilize a halocarbon compound or ammonia as the refrigerant. An internal combustion engine, turbine, or electric motor supplies the power to drive a centrifugal or positive displacement compressor.
Refrigeration, or cooling, occurs when the liquid refrigerant absorbs heat by evaporation, generally at low temperature and pressure. When the refrigerant condenses, it releases heat to any available cooling medium, usually water or air.
SINGLE-STAGE REFRIGERATION CYCLE
The basic refrigeration cycle used for single-stage vapor compression has four components in the system. They are the compressor, condenser, metering device, and evaporator. Low-pressure liquid refrigerant in an evaporator extracts heat from the fluid being cooled and evaporates. The low-pressure vapor is then compressed to a pressure at which the refrigerant vapor can be condensed by the cooling media available. The vapor then flows to the condenser, where it is cooled and condensed. The liquid refrigerant flows from the condenser to a metering device, where its pressure is reduced to that of the evaporator. The cycle is thus completed.
In industrial or commercial air conditioning systems, the heat is usually rejected to water. Once-through cooling may be used, but municipal restrictions and water costs generally dictate recirculation and evaporative cooling processes.
Evaporative condensers or cooling towers are normally used for evaporative cooling. A spray pond may be used as an alternative. Recirculation of the water in a cooling system reduces the makeup water requirement to less than 3% of the water that would be needed for once-through cooling.
Cooling capacity is measured in tons of refrigeration. A ton of refrigeration is defined as the capacity to remove heat at a rate of 12,000 Btu/hr at the evaporator or chiller.
An absorption refrigeration system that removes 12,000 Btu/hr (does 1 ton of air conditioning) requires heat energy input of approximately 18,000 Btu/hr to drive the absorption process. This means that the heat rejection at the cooling tower approximates 30,000 Btu/hr per ton of refrigeration. With a 15°F (8°C) temperature drop across the tower, the heat rejection of an absorption system requires circulation of approximately 4 gpm of water per ton of air conditioning. Evaporation of the recirculating water occurs at a rate of approximately 3.7 gph per ton.
Other than the solution and refrigerant pumps, there are no moving parts in an absorption system. Although this is an economical design advantage, the cost of producing the necessary low-pressure steam or high-temperature hot water (HTHW) must also be considered.
Compression systems also impose an additional heat load. This is due to the energy required to compress low-pressure, low-temperature refrigerant gas from the evaporator and deliver it to the condenser at a higher pressure. The compressor energy input is approximately 3,000 Btu/hr per ton of refrigeration. Accordingly, normal heat rejection in a compression system approximates 15,000 Btu/hr per ton of refrigeration, requiring evaporation of about 2 gal/hr of cooling water.
Compression refrigeration systems require a cooling water circulation rate of approximately 3 gpm per ton of refrigeration, with a 10°F temperature drop across the cooling tower.
The major energy consumer in a compression refrigeration system is the compressor, which is designed to operate at a certain head pressure for a given load. This pressure equals the refrigerant pressure in the condenser. The term "high head pressure" refers to condenser pressure that is higher than it should be at a specific load condition.
High head pressure can be costly in two ways. First, it presents the danger of a system shutdown; a safety control will stop the compressor motor when the safe maximum head pressure is exceeded in the compressor. Second, an increase in power consumption results when a compressor operates at greater than design head pressure.
Fouled condenser tubes are a common cause of high head pressures. Fouling increases the resistance to heat transfer from the refrigerant to the cooling water. In order to maintain the same heat transfer rate, the temperature of the refrigerant must be increased. The compressor fulfills this need by increasing the pressure at which the refrigerant is condensed. With a centrifugal chiller, a 1°F increase in condensing temperature increases compressor energy consumption by approximately 1.7%.
Fouling and the formation of scale in absorption systems also reduce operating efficiency. Because the highest water temperatures exist in the condenser, deposition first occurs in this unit. Under extreme conditions, scale formation can also occur in the absorber.
Deposition in the condenser imposes a higher back-pressure on the generator, so that increased steam or HTHW is required to liberate the refrigerant from the absorbent. The result is an increase in refrigerant vapor pressure and a greater temperature differential between the condensing water vapor and the cooling water. Although this compensates for the resistance to heat flow, more energy is required to provide the increased heat input.
If water conditions are severe enough to cause deposition in the absorber, less refrigerant is removed by the absorber, and cooling capacity is reduced. The reduction in refrigerant circulation diminishes the ability of the equipment to satisfy cooling requirements.
If the absorption rate in the absorber is reduced while the absorbent is heated above the normal temperature in the generator, the danger of over-concentrating the brine solution also exists. This over-concentration can cause crystallization of the brine, leading to a system shutdown.
Fouling and scale formation waste energy and can ultimately cause unscheduled system shutdown. Effective water treatment can minimize the possibility of high head pressure and excessive steam consumption caused by condenser deposition.
Corrosion can cause problems in either the open recirculating or chilled water circuits. When corrosion is not properly controlled, the resulting corrosion products inhibit heat transfer, increasing energy consumption in the same manner as fouling and scale formation. Unchecked corrosion can cause heat exchanger leaks and catastrophic system failures.
In any cooling application, attention to cooling tower operation is important. Proper tower maintenance maximizes cooling efficiency, or ability to reject heat. This is critical for continuously running refrigeration machinery at full load conditions.
For best performance, the cooling tower fill should be kept clean and protected from deterioration. The water distribution system must provide uniform wetting of the fill for optimal air-water contact.
Other components, such as drift eliminators, fill supports, regulating valves, distribution decks, and tower fans, should be kept clean to maintain efficient heat rejection. Inefficient cooling or heat rejection increases the temperature of the water in the cooling tower sump and, consequently, that of the water sent to the condenser. This makes it necessary to condense the refrigerant at a higher temperature (absorption) or higher temperature and pressure (compression) to reject heat at the same rate into the warmer water. This increases the amount of energy (steam, hot water, electricity) required to operate the system.
OPEN RECIRCULATING COOLING WATER PROBLEMS
The water in open cooling systems is subject to problems of scale, corrosion, slime, and algae.
Scale
As water evaporates in a cooling tower or an evaporative condenser, pure vapor is lost and dissolved solids concentrate in the remaining water. If this concentration cycle is allowed to continue, the solubilities of various solids will eventually be exceeded. The solids will then deposit in the form of scale on hotter surfaces, such as condenser tubes. The deposit is usually calcium carbonate. Calcium sulfate, silica, and iron deposition may also occur, depending on the minerals contained in the water. Deposition inhibits heat transfer and reduces energy efficiency.
Deposition is prevented by threshold inhibitors that increase the apparent solubility of the dissolved minerals. Therefore, they do not precipitate and are removed by blowdown. Blowdown is automatically replaced by fresh water.
The ratio of dissolved solids in the circulating water to that in the makeup water is called "cycles of concentration." With correct treatment, cycles of concentration can be increased so that less makeup water and consequently less chemicals are required (Table 34-1).
| Daily Water Requirements (gal) | Yearly Water Costs | |||
| Tower size tons of refrigeration |
2 cycles | 5 cycles | 2 cycles | 5 cycles |
| 250 | 10,800 | 6,750 | $972 | $608 |
| 600 | 25,920 | 16,200 | $2333 | $1458 |
| 3000 | 129,600 | 81,000 | $11,664 | $7290 |
The cooling capacity of a tower is affected by how finely the water is atomized into droplets. Smaller droplets lose more heat to the atmosphere; however, more of the smaller droplets are carried away by the air drawn through the tower. This "windage loss," or "drift loss," becomes part of the total blowdown from the system. Windage loss is approximately 0.1 to 0.3% of the water circulation rate.
Windage can have undesirable effects, such as the staining of buildings and the spotting and deterioration of car finishes. These problems are caused by the dissolved solids in the circulating water, which evaporate to dryness as water droplets fall on surfaces. Because water treatment chemicals produce only a slight increase in the dissolved solids content of the water, they usually do not contribute significantly to spotting problems.
Continuous blowdown, or bleed-off, is adequate for scale control in some cooling systems. The importance of continuous blowdown, as opposed to periodic complete draining, cannot be overemphasized. The volume of water in most cooling systems is small compared to the amount of water evaporated. Therefore, excess solids concentrations can develop in a short period of time. Continuous blowdown prevents excessive solids concentrations from developing in the tower water.
In order to maintain solids in solution, water that is high in alkalinity and hardness may require the feed of sulfuric acid or an acid salt in addition to blowdown. Acid feed requires careful handling and control and should only be used where the blowdown rate would otherwise be excessive.
Sodium zeolite softening of the makeup water is also an effective way to control scale. However, this process does not decrease the alkalinity of the tower water. Because the resulting low-hardness, high-alkalinity, high-pH water is particularly aggressive to copper alloys, it may necessitate acid feed in addition to softening. Also, carbon steel corrosion control is more difficult with softened water than with hard water.
Polyphosphates are of some value for scale control but must be applied cautiously, because hydrolysis of the polyphosphate results in the formation of orthophosphate ions. If this process is not properly controlled, calcium phosphate deposits may result. Chemicals are now available that inhibit scale formation without this undesirable side effect. Therefore, polyphosphates are now used primarily for corrosion inhibition.
A treatment which controls calcium carbonate particle growth and prevents deposition can permit a reasonable blowdown rate and eliminate the need for pH depression with acid. Phosphonates are particularly useful as threshold inhibitors of scale formation and as iron oxide dispersants. Certain low-molecular-weight polymers also have the ability to control calcium carbonate scale formation.
Suspended solids (airborne dust and debris from the air passing through the cooling tower) contribute to general fouling and can aggravate scale formation. The deposits may also cause localized under-deposit corrosion.
Fouling of heat transfer surfaces has an insulating effect that reduces the energy efficiency of the process. Failure to control scale formation also reduces the rate of heat transfer. Accordingly, a properly designed treatment program must include polymeric dispersants and scale control agents to minimize general fouling and inhibit scale formation.
Corrosion
Water in an open recirculating cooling system is corrosive because it is saturated with oxygen. Systems in urban areas often pick up acidic gases from the air that can be beneficial in scale reduction. However, excessive gas absorption can result in severely corrosive water.
Chromate-based corrosion inhibitors are very effective, but their use is now prohibited in comfort cooling towers. The most commonly used corrosion inhibitors are phosphate, molybdate, zinc, polyphosphate, silicate, and organic-based treatments. These inhibitors can be applied in low or alkaline pH treatment ranges.
At low pH, a high phosphate level is used to promote the passivation of steel. At high pH, a combination of various corrosion inhibitors and deposit control agents is used. These programs use organic inhibitors in combination with zinc, phosphate, or molybdate. Where these are environmentally unacceptable, silicates may be used at an alkaline pH. This type of inhibitor program also includes deposit control agents. However, silica concentration must be controlled to prevent deposition of silicate, which forms a hard and persistent scale.
Azoles, functioning as copper corrosion inhibitors, are used in most programs to improve the corrosion protection of copper and to minimize pitting of ferrous metals.
Because the heat load on many cooling tower systems varies with changing weather conditions, water evaporation rates tend to be irregular. As a result, cooling system protection can be less than desired or expected under conditions of wide load fluctuation. Automated water treatment control equipment substantially improves treatment results in systems that operate under these conditions.
Slime and Algae
Many types of antimicrobials are available for control of algae and biological slime in open cooling systems. Nonoxidizing organic materials (such as quaternary ammonium salts, other organic nitrogen compounds, and organosulfur compounds) are frequently used. Some antimicrobials can be detoxified before discharge into the environment. Microbiological programs often employ a combination of nonoxidizing and oxidizing chemicals. Oxidizing chemicals include chlorine, hypochlorites, organic chlorine donors, and bromine compounds. Chlorine gas requires chlorination equipment and controls, which are not practical for most air conditioning systems. Chlorine and hypochlorites must be applied carefully, because excessive chlorine will increase corrosion and may contribute to deterioration of cooling tower wood and reduction of heat transfer efficiency. For more information on microbiological problems and antimicrobial use in cooling systems, see Chapter 26.
Air washers are spray chambers in which air is conditioned by direct contact with water. The chilled water is contained in an open system, or circulated from a closed system .
Air washers remove dust, smoke, and odors from the air. Additionally, the return air from a manufacturing process may contain unique contaminants that must be removed. Process contaminants include fiber and oil in textile plants, tobacco dust in tobacco plants, and sizing material in cloth weaving plants.
Filters remove particulate matter from the air before it passes through the spray section. Eliminator blades prevent mist or water droplets from leaving the unit with the air. In addition to cleaning, air washers usually perform other functions. Air temperature and humidity can be controlled through adjustment of the spray water temperature.
When air must be humidified in winter, some water is evaporated. This increases the solids concentration in the remaining liquid. Generally, scale does not form, because the temperature of the water is relatively low. If the spray water temperature is below the dew point of the entering air, the air is dehumidified. In summer, dehumidification involves condensing water vapor from the air, diluting the solids in the recirculating water, and causing water to overflow from the bottom sump or pan of the air washer.
Corrosion can develop in air washers as it does in recirculating cooling water systems. The spray water is saturated with atmospheric oxygen and, when present, acidic airborne contaminants lower the pH and contribute to its corrosivity. Accordingly, the use of a corrosion inhibitor is important in air washer treatment.
Cleanliness of the air washer helps to prevent objectionable odors in the air. The volume of air in relation to the water circulation rate in air washers is much greater than it is in cooling towers. Therefore, the tendency to accumulate sludge is much greater. Sludge can cause localized corrosion or promote biological activity that produces odors. Therefore, dispersants and/or surfactants are an integral part of a water treatment program for air washers.
The air being washed also contains numerous microorganisms and materials that will feed bacteria. Therefore, biological slime is a significant problem in air washers. Nonoxidizing chemicals are used for microbiological control. However, undesirable odors may result from this treatment.
If the air washer must be sterilized, the air flow is stopped and a solution of oxidizing or nonoxidizing antimicrobial is circulated through the washer. The unit must then be hosed until the material loosened by the treatment is thoroughly flushed from the bottom of the air washer.
Closed systems are not subject to scale formation except when hard makeup water must be used. Many closed systems use zeolite-softened water or condensate as makeup to prevent scale problems.
In closed systems, the oxygen concentration is lower than that of aerated systems. Therefore, the potential for corrosion is much lower. However, some corrosion exists, and loose corrosion products can cause fouling of piping, automatic valves, and vents.
Theoretically, closed water systems should not require corrosion inhibitors. Any oxygen introduced with the initial makeup water should soon be depleted by oxidation of system metals, after which corrosion should no longer occur. However, closed systems usually lose enough water and leak enough air to require corrosion protection.
The inhibitors most commonly used are molybdate, silicate, or nitrite based. The use of chromates may be restricted because of regulations that classify them as carcinogens. The amount of inhibitor needed depends on the system water temperature and its metallurgy. Closed systems usually require little additional treatment after the initial charge. Therefore, relatively high treatment levels can be used to provide a greater margin of safety at relatively low cost.
Sulfite-based programs are also used to control corrosion. Unlike the other inhibitors, a corrosion inhibitor film is not established; the sulfite prevents oxygen corrosion by reacting with and removing dissolved oxygen. An alkaline pH is maintained to prevent acidic corrosion. In the case of air leaks, sulfite requirements are not proportional to water loss and can be very high. High sulfite consumption adds to the dissolved solids content of the circulating water and increases the cost of treatment. Therefore, air in-leakage should be minimized.
Insulating couplings are used in closed systems to control galvanic corrosion. These couplings are mostly phenolic resins, which may be attacked at high pH.
Weather changes cause solids concentration changes in open cooling water systems and particularly in air washers. Air conditioning system design does not always properly address water treatment needs. Often, water sump volumes are reduced in cooling tower designs to minimize system weight. This results in a lower ratio between volume and circulation rate, which causes a more rapid change in water solids concentration with variations in load. Also, low-capacity water pans are used in evaporative condensers and air washers in order to reduce space and weight.
A water treatment program can be complicated by any of the following factors:
- cooling towers near smokestacks can pick up airborne dirt and acidic gases
- cooling towers are often installed and operated in such a way that considerable water overflows from the system on shutdown
- additional makeup water may be required during hot weather to reduce water temperature
For open systems, an effective and efficient treatment program includes continuous blowdown, continuous feed of corrosion inhibitor and dispersant, and daily water testing. Systems should not be treated and controlled solely on the basis of weekly tests. Additional chemical treatment may be needed to ensure adequate protection.
The importance of beginning chemical treatment promptly cannot be overemphasized. New installations will have mill scale on metal surfaces and will contain oil, pipe compound, brazing and welding scale, and construction debris. Systems previously operated without water treatment (or with ineffective water treatment) contain corrosion products which can slough off when protective treatment is started. Such materials can impede water flow, cause fouling, and increase galvanic corrosion potential. Suspended solids may cause automatic valves and controls to malfunction, and can shorten the life of mechanical seals on the pumps.
Such systems should be thoroughly cleaned (mechanically and chemically), drained, and flushed. Cleaning agents commonly used are organic phosphates, polyphosphates, synthetic detergents, dispersants, and combinations of these materials. The permanent protective treatment should start immediately after cleaning because clean metal surfaces in the system are particularly vulnerable to corrosion.
Air conditioning systems that do not operate all year should be protected properly during idle periods. Open cooling water systems should be drained completely. Condensers should be opened and inspected at the end of every air conditioning season. The basins of cooling towers or air washers should be thoroughly cleaned and flushed.
If the system is stored dry, the condenser should be closed tightly after it is thoroughly dried. Ideally, it should then be filled with nitrogen and sealed. If water is not removed from an idle system, additional protection is required to offset the increased corrosion potential. A higher concentration of a corrosion inhibitor suitable for long-term storage must be used.
If closed system water temperatures will be at or below freezing, antifreeze must be added for protection. When ethylene glycol antifreeze is used, chromate-treated water must be drained from the system because these materials are not compatible. However, chromate is compatible with methanol, calcium chloride, and sodium chloride brines. Molybdate, nitrite, and silicate-based inhibitors may be used with any of these antifreeze solutions.
Eliminator sections of cooling towers can collect salt deposits as a result of partial or intermittent wetting. Because sufficient amounts of the treated water do not reach the eliminator sections during operation, treatment chemicals added to the water cannot be expected to provide protection. Salts, dirt, and debris also accumulate on the eliminators and screens of air washers and evaporative condensers. Such areas should be hosed down at regular intervals.
Where appreciable dirt has collected, mechanical cleaning is needed. In industrial plant environments, mechanical cleaning may be required several times during an air conditioning season.
Figure 34-1. Good water treatment practice and scheduled maintenance can help prevent unexpected shutdown of air conditioning equipment.

Figure 34-2. Absorption refrigeration system.
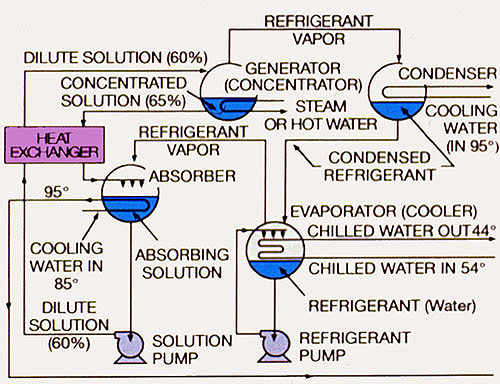
Figure 34-3. Compression refrigeration system.
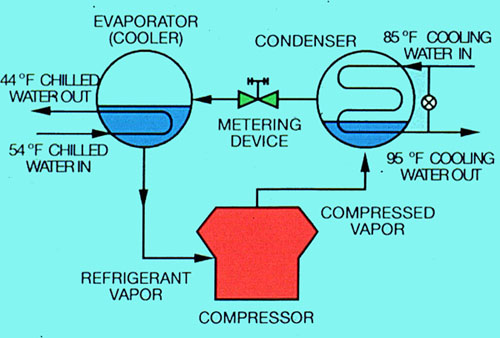
Figure 34-4. A fouled condenser can increase head pressure and waste energy.

Figure 34-5. Failure to control biological slime reduces heat transfer.

Figure 34-6 Air washer with open chilled water system.
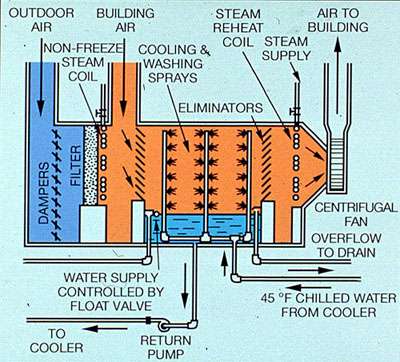
Figure 34-7 Air washer with closed chilled water system.